POLLUTION
Item
- Title
- POLLUTION
- extracted text
-
LEGEHO
Soi^mentSon^oL^^^
r?PPS1
Ucf-en Samp(-> l o^K^3
© CMl.
Water Sarnpl.- locate™
AWS
''
L1<WA
9t
I
i >
A
uK
ri
;
i
'V:
r<
:uy
X
L?V
x 19
/OSB®';
r/
• .
.
4
z• '• . //
/ /
1/JS5
<jl.O
A
)
n
.
/<•
z
A
•
•
. ' <-cJL
M
fm?
Hil
F A ro A
$US2
USW1
IMS®
;^)usi
fr) CC1
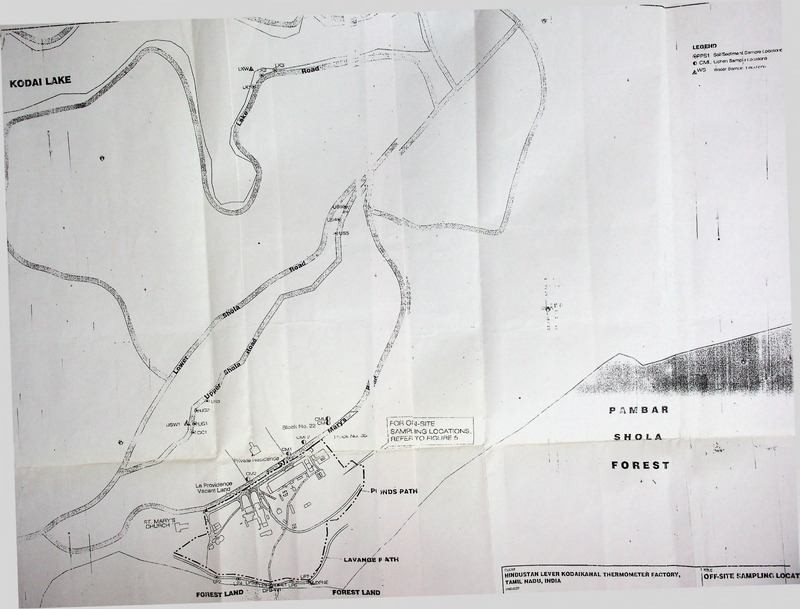
FOR ON-SITE
iCK NO. 3b
r? .
I sampling locations.
j REFER TO FIGURE 5
)
S H O LA
srr
FOREST
A
La Providence
Vacant tape
)NDS PATH
ST MARY’S
church '
I
1
LAVANGEP ATH
CLICNT
HINDUSTAN LEVER KODAIKANAL THERMOMETER FACTORY
TAMIL NADU, INDIA
LP7
dfne
FOREST LANO
FOREST LAND
f-noJCCT
1 im.c
|OFF-SITE SAMPLING L
': T
• 4» ’ ■*/<
«‘ <•' 5^
’*
4
'■'i-r
$
•
ME%rx-.-z-/ -<cJgrx?4-»P»M
fj?
^?i;fzenya ..v
U^r^njgaf
uA^i AS
CCSIQf-ro
DSW
DRAWN:
HO
« ***—lz-u
I status
DHeC^tE_________
1903 a
=POJCC-
■»
^ika;.
49032^002_353
A5032_002_00<
A
o
WrS6w
5C0
metres
J V,
>S$3
-^T'£iccjHV<,’jZ’(qfc*5
•; c •. J ; r • v
*‘
; I?3
o
so
^FERA\lBUKAblAL{
.•i
Q?
) • • .• 2229
-; i^c^^i^cide.-.F^pint'/:
LEGEND
rax*
□self's1
*?x
TOd
Jcru^.
, Fed'jungle
Temple
ioBsfis
?7
5
I
I Hindustan lever kodaikanal
! THERMOMETER EA
NADU, INDIA
ENVIRONMENTAL SITE AND
RISK ASSESSMENT J-G
MERCURY
TOPOGRAPHY MAP
M’
'■4.W
*
•
a
1 1•
1
J CES»Gr<O
DSW
HC
1 CRAV.n
1 CHCCS^ED
block No. 3b
f
J
STATUS
‘’-’OjSCT
49CWZ_CXXZ_353
>-Dr<>.i
49CC2 OO? OO1
HE/lSM-i
A
20
0
■
J
APPOOLTO
OATE
j
-tKAL
40
metres
<
Existing Building Numbers
FOOT PATH
i
P nvai
3c
' \3a
w
8 •
i
'*jAREA A j
I ch
J
9a\9
\
1
PONDS PATH.
1
— Site Drainage Stream
12 V.
f-j
\16
23 x
21
LAVAN GE PATH
250
>AREA G
1. Mam Factor/
2. Old Admin Block
3. Generator
3b. Switch Room
4. LPG Storage Yard
5. Diesel Storage Yard
5. Compressor Cooling To-'/er .
Collection Tank Sumo
3. C j-Hecton Treatment Se~ r.~
9. Aeration Tank
9a. TW Collection Sump
10. Crusher/Recovery Area
' 1. Existing Well & Pump House
12. Rain Water Collection Sumo
12a. Pump Shed
13. Sump
14. Rain Water Collection Su
15. Cooling Water
16. Eastern Spring Tank
17. Distillation Plant
13. Sludge Storage Arsa
19. Septic Tank
20. Domestic Sewage Tieairnen: Piar;
21. Waste Glass Store
22. Incinerator (Not in use.)
23. Sump
27. Canteen Water ColleciK>7
28. Kitchen (Fire Wood Fuel)
30. Compressor Room
31. Servant Quarters
32. Staff
33. Staff Quarters
34. Transformer Yard
35. Security Room
36. Spring Tank
LEGEND
o SA2 Additional Soil Sanipte uc-caDor^
.•.PPS1 Soil/Sediment Samoa Lscatcrs
C CVLUchen Sample Locations
AREA D
*v5
Water Sample Locaoons
Proposed Remsdieocri Areas
CLENT
?---
FOREST LAND
HINDUSTAN LEVER KO DAI KAMAL
THERMOMETER FACTORY, TAMIL
NADU, INDIA
S1 «
7m
project’
I
V
ENVIRONMENTAL SITE AND
RISK ASSESSMENT FOR
MERCURY
s
*
*
$
!
Zm
i
FOREST LANE
I
1
I
ISITE LAYOUT AND SAMPLE f
I location plan
;
1 dm
r
S11 o -
AdditionalSampiingLocations Jn Area c
J—«-«a
a Mercury
Thermemeter
Keundup
The Problem with
Mercury Thermometers
Mercury' thermometers have been used
for decades as a first step to care for
someone who isn’t feeling well.
Ironically, mercury fever thermometers
can be a risk to the health of families
and communities. Public health offi
cials report over 15,000 calls a year to
poison control centers about broken
mercury' thermometers. A thermometer
contains about 0.7 to 1.5 grams of mer
cury. Fever thermometers are one of
the largest single sources of mercury'
discarded annually in municipal solid
waste, estimated at 17 tons of mercury.
Why Hold a Mercury
Thermometer Exchange??
Exchanges are easy to hold with big
payoffs. Depending upon how big or
elaborate you envision your event,
coordinating an exchange is relatively
easy. The benefits of an exchange are
numerous:
a When given the information about
the hazards of mercury' thermome
ters, people are more than willing
to find a safe place to get rid of
them.
°
Providing a free non-mercury alter
native is a big bonus; people are
always thankful to get free things,
especially when the associated ben
efits are so positive.
Q
The public image and media oppor
tunities should not be overlooked.
This is a win-win situation for
everyone and the public will be
receptive to that message.
Planning your Exchange
The success of a roundup depends on
the successful promotion of the event.
If people do not know about the event,
not only will they not turn in their
thermometer, but the opportunity' for
education on the health and environ
mental impacts of mercury' will be lost.
A hospital exchange is relatively simple
to undertake. Primarily, this is because
your audience is easily defined. You
have a few straight-forward means to
promote the event. Promotion is there
fore simple and inexpensive. In addi
tion, the audience is a known quantity'.
Based on the number of employees, a
simple formula can be used to estimate
the number of exchange thermometers
needed for purchase and disposal.
Experience has shown that 15%-20%
of hospital employees will bring in their
home thermometers.
Choosing a Non-Mercury
Thermometer
While there are a variety of mercuryfree thermometers available in the
market place, there are primarily two
types that fall within the budget of an
exchange. These alternatives are the
geratherm thermometer, and the digital
thermometer. Your purchasing depart
ment can easily get prices on these two
alternatives.
Funding
Before you go looking for funding it is
important to know what you are asking
for. Is it money for thermometers, or
other in-kind support? The simplest
exchange requires thermometers, dis
posal, and perhaps some money for
printed promotional materials.
Compared to many programs, the
funding budget for a thermometer
roundup is rather small.
High profile events will typically
require a higher budget to fund food,
receptions, etc. Including these
niceties can mean adding the task of
intensive fundraising to the work of
organizing an event.
Mercury Thermometer
Disposal
Those helping with the exchange and
those turning in their thermometer
will want to know the eventual fate of
the mercury* in the thermometers.
Currently, the mercury in fever ther
mometers and other mercury-containing devices is recycled using a process
called “roast, retort and distillation.”
Basically, the mercury-containing
items are crushed, and heated so that
the mercury’ evaporates and is thus
separated from the glass and other
debris. The gaseous mercury’ is then
retorted or condensed back to a liquid
state. The liquid mercury' is then dis
tilled to remove impurities and can be
used again in new mercury-containing
products.
o
o
Publicity
n
Fliers
■ Table tents - dining room, staff
lounges
°
Newsletters
a
E-mail announcements (the day
before event, post an automatic
announcement - “don’t forget your
mercury’ thermometer tomorrow!”)
H
Announcement in payroll checks
Safety and Environmental
Logistics
It is important to make sure that in all
promotional materials participants are
told to bring in thermometers in rigid
containers. This can help protect
against problems should the ther
mometer break on the way to the
exchange event.
Work with workplace or state safety or
hazardous materials specialists during
the event planning process to ensure
regulatory' and compliance issues are
being considered. If you are going to
transport the collected thermometers
to the disposal facility' it is important
to ensure that transport and labeling
regulations are being followed.
Mercury debris treated for reclamation
is considered a “universal waste”, but
contact your state’s hazardous materi
als section to ensure you will be in
compliance with your state’s environ
mental requirements.
Someone with mercury spill cleanup
training should be on hand at the
event with mercury’ spill equipment.
Reception
A workplace reception can be a great
way to help promote the exchange, but
at the same time has the potential to
use a lot of planning time. In the hos
pital setting, by virtue of medical pro
fession involvement, a reception can
help draw attention to mercury' as a
public health issue. At a reception it is
useful to have a display on mercury,
mercury-free alternatives in the home
and workplace, and mercury' pollution
prevention literature.
Options for speakers include physi
cians or clinicians that can speak to
the health hazards of mercury, state or
federal speakers addressing the status
of mercury legislation, and local envi
ronmental organization representatives
and workplace staff on what that
organization is doing to address mercu
ry reduction/elimination.
Educational Opportunities
Before the event, collect enough edu
cational materials to distribute. In
addition to the mercury' publications
included in this resource kit, you may
want to provide:
□
Your state fish advisories
□
List of other mercury-containing
items in the home
□
Local and state contact informa
tion about disposal options for
other mercury-containing house
hold items
Location and Schedule of
the Exchange
It is important to time your exchange
so that is convenient for those partici- |
paling in the exchange.
In a workplace with shift workers, try'
at a minimum to schedule the
exchange over one shift change. It is
easy for exchange participants to trade
in their thermometer at the beginning
or end of their shift. If possible try to
hold the exchange over at least a twohour minimum. The longer rhe event,
the greater the chance the internal
word of mouth will remind workplace
staff of the event.
Holding the event in a popular com
munity meeting area will also help the
success of the exchange. Typically, the
most successful meeting place is the
cafeteria. Setting up the “exchange
(
table” outside the cafeteria doors will
guarantee a steady stream of people. In
many hospitals there is a shift change
at the lunch hour. Accounting for
location and timing will help the
exchange tremendously.
Other Considerations
Either due to good promotion or a
small budget you should also be pre
pared with a contingency plan should
you run out of thermometers. Will you
offer a voucher that the participant
can redeem in the future, will you turn
them away, or do you promote the
exchange of free thermometers only
“while quantities last?”
•
You can involve your hospital pharma
cy and local drugstores by asking them
to provide discount vouchers for mer
cury-free thermometers if your supply
runs out. At the same time, you can
ask them to no longer sell mercury
thermometers. If you are organizing an
exchange in a hospital, you can pre
sent embarassing questions by assuring
people that the hospital pharmacy has
ended the sale of mercury' thermome
ters.
3. When the tray “fills up,” wrap the
stack of unbroken thermometers in
bubble wrap, secure with rubber
band and place in a collection con
tainer. The collection container
should be labeled “Mercury'
Thermometers” and any rigid con
tainer that has a lid will work.
(Five gallon containers used in
food service or for dry-wall spackle
work well.) Participants should not
reach in or place their thermome
ters directly into the container.
4. Broken thermometers should be
placed directly into the collection
container without removing them
from their rigid plastic container.
For more detailed information,
see HCWH s 12-page booklet How to
Plan and Hold a Mercury Thermometer
Exchange.
Collection Procedure
1. Participants remove unbroken
thermometer from rigid container.
Dispose of container in a recycling
bin and place thermometer on a
piece of bubble wrap spread on a
tray.
2. Keep track of the number of ther
mometers collected and the num
ber of families participating. A flip
chart may be used to visually show
progress throughout the exchange
event.
3
Battery
Round-Ups:
Get Charged!
The Problem With Batteries
Mercury-Containing Batteries
Many different types of batteries are in
use in hospitals. Pagers, infusion
pumps, fetal monitors, portable EKG
monitors, flashlights, smoke detectors,
hearing aids, and portable generators
are just a small sampling of devices
that use batteries in hospitals. Several
types of batteries contain mercury and
may also contain other heavy metals
such as lead and cadmium.
■
Many hospitals have battery-recycling
programs for a portion of their batter
ies. Unfortunately, there is consider
able confusion on proper management
methods for batteries. This confusion
can lead to poor capture rates, and
improper disposal of batteries into red
bag waste.
Common uses: pacemakers, defibril
lators, fetal monitors, heart moni
tors, pagers, telemetry devices, tem
perature alarms and blood analyzers
Recycling/disposal options: recycle to
reclaim mercury
°
A battery round-up is an excellent way
to provide education on the hazards
associated with batteries, and on prop
er battery management to hospital staff
and their families. It is also an excel
lent way to initiate, or improve upon,
an ongoing, comprehensive battery col
lection program. Finally, they are an
excellent follow-up to a mercury ther
mometer collection program.
Within a hospital, a number of different
types of batteries are utilized. Special
care should be taken to separate each
type individually, as they are disposed
of in different ways, depending on their
content. Batteries should not be incin
erated. The battery types to look for in
your facility include:
Alkaline and Carbon-zinc (nine
volt, D, C, AA, AAA, alkaline but
ton) Alkaline and carbon-zinc bat
teries contain chromium and zinc,
and older ones (pre-1996) may con
tain mercury. All imported batteries
(even new) are likely to contain
mercury (except those manufactured
in Western Europe and Japan, which
may contain trace levels). These are
classified as non-hazardous.
Common uses: pumps, diagnostic
equipment, defibrillators, oto
scopes, opthalmoscopes, dictation
machine, pen lights, glucometers,
flash lights and telemetry devices
What is a Battery Round-up?
A battery round-up is a permanent
hospital-wide battery collection and
recycling program for employees and
their family members. All non-mercury
containing batteries are collected
for proper disposal (they will not be
incinerated) and all mercury-containing batteries are recycled.
Mercuric-oxide (button, some
cylindrical, and rectangular)
Mercuric-oxide batteries contain the
highest percentage of mercury, and
are classified as hazardous waste.
Businesses and institutions are
required to manage these hazardous
materials through recycling or haz
ardous waste treatment/disposal.
Recycling/disposal options: recycle
older alkalines to reclaim mercury;
recycle newer alkalines to reclaim
zinc, or dispose of in a landfill or
treat as hazardous waste
Non-Mercury Containing
Batteries
The following batteries are classified as
hazardous waste. Businesses and insti
tutions are required to manage these
hazardous materials through recycling
or hazardous waste treatment/disposal.
■
Lead-acid (button, some cylindrical
and rectangular) Lead-acid batter
ies contain lead. Some are
rechargeable.
Common uses: wheelchairs, portable
generators
Recycling/disposal options: recycle to
reclaim lead, or treat as hazardous
waste
■
Nickel-cadmium (9 volt, C, D,
AA, AAA, battery packs) Nickel
cadmium batteries contain high
levels of nickel and cadmium.
They are labeled as rechargeable.
Common uses: emergency lighting,
portable communication devices
and medical equipment backup
Recycling/disposal options: recycle to
reclaim nickel and cadmium, or
treat as hazardous waste
Silver-cadmium (9 volt, C, D, AA,
AAA, battery packs) Silver-cadmi
um batteries contain silver and
cadmium. These batteries are
rechargeable.
Laboratory. Other important stake
holders to include are: State
Hazardous Waste or Pollution Control
Agencies and your hospital recycling
contractor(s). Anticipate six months to
plan your battery round-up.
Important committees to include in
the planning process are:
n Fundraising - to cover printing
costs for posters, tent cards, adver
tising and the reception;
n
Event Planning - a high visibility
event and reception for employees
and family members that will mark
the beginning of a permanent hos
pital-wide ongoing battery collec
tion and recycling program;
a
Publicity - internal public informa
tion planning ( posters, email
alerts, tent cards for tables,
newsletters, etc.) and external
media communications; and
°
Education - responsible for devel
opment of educational pieces for
distribution to hospital workers and
their families about battery recy
cling, including types of batteries
used in health care, examples of
their use, and mercury content.
Common uses: medical electronics
Recychng/disposal options: recycle to
reclaim silver and cadmium, or
treat as hazardous waste
H
Small sealed lead-acid flat plates
(gum packs, pack configurations)
Small sealed lead-acid flat plates
contain high levels of lead. They
are labeled and are rechargeable.
Common uses: emergency lighting,
portable communication devices,
medical equipment backup and lap
top computers
Recycling/disposal options: recycle to
reclaim lead, or treat as hazardous
waste
Planning
Such a program may seem like a big
undertaking, but with proper planning
a battery round-up provides for good
public relations, employee morale, and
potential savings from the elimination
of battery disposal in red bag waste.
Important stakeholders to involve in a
planning team include: hospital depart
ment staff from Safety, Facilities,
Community Relations,
Communications, Purchasing and
Resources
Recycling America’s Rechargeable Batteries. The
Plan. Rechargeable Battery Recycling Corp
Reducing Mercury' Use in Health Care
“Greening Hospitals” HCWH
Mercury’ Disposal Options for Region 1 US EPA
June 1999 by Rebecca Herman, contractor
Mercury' Pollution Prevention in Healthcare: A
Prescription for Success by Guy Williams
Pollution Prevention for Health Care Facilities
by Hollie Shaner.
11th International Seminar on Battery' Waste
Management, Conference Literature
Florida Educational Seminars, Inc. (Sponsored
by the Battery Industry)
Implementation oi the Mercury-Containmg &.
Rechargeable Battery' Management Act
(EPA530-K-97-009)
Used Dry’ Cell Batteries: Is a Collection Program
Right for Your Community US EPA EPA 53O-K92-006
Universal Waste Rule US EPA EPA53O-F-95-O25
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
inlo@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at w;vw.noharm.org.
I^SoyjNKi.
The rCF certification mirk and term arc the sole property or the Chk-nnc Free
Products A'uKi.ii.cn and air only u <d by authorized and ieitif.ed u»cr*
PVC, and
[HJeatth Care
Institutions
What is dioxin?
Dioxin is the name given to a group of
persistent, very toxic chemicals. The
group includes chlorinated dibenzo
dioxins, the most toxic of which is
2,3,7,8 -tetrachlorodibenzo-p-dioxin
(TCDD), and chlorinated dibenzo
furans. The group also includes related
compounds which are structurally simi
lar and are dioxin-like in their activity.
The toxicity of these compounds is
measured against TCDD using “toxic
equivalents,” which assign a fractional
potency to each dioxin. Dioxins,
defined here to include dioxins and
furans, have equivalence factors
assigned to them. The EPA has not
assigned equivalence factors for brominated dioxins, brominated furans,
brominated biphenyls and PCBs,
although it is believed each group
includes some dioxin-like compounds.
Dioxins and related compounds are
highly persistent in the environment
and in living organisms. They are
bioaccumulative and fat-soluble. Their
concentrations increase as they bio
magnify up the food chain.
What are the
hazards of dioxin?
Dioxins are extremely toxic and potent
environmental contaminants. They
modulate and disrupt growth factors,
hormones, enzymes, and developmen
tal processes. In animals, dioxin causes
cancer in multiple organ systems,
sometimes at exposure levels as low as
nanograms per kilogram of body
weight. Prenatal exposure to dioxin in
rodents substantially increases the risk
of breast cancer later in life.1 Human
epidemiological studies conclude that
dioxin causes cancer in humans as
well.2 A draft report by the EPA esti
mates that as many as one in 1,000 of
the most highly exposed people in the
general population are at risk of devel
oping cancer because of dioxin.
Dioxin also has widespread effects on
reproduction and development, as
shown in animal and human studies.
Tiny doses in the range of nanograms
(one thousandth of one millionth of a
gram) to micrograms (one millionth of
a gram) per kilogram of body weight of
dioxin can cause harm. Exposure to
these levels on a single day during
pregnancy cause permanent disruption
of male sexual development in rodents,
including delayed testicular descent,
lower sperm counts, and feminized sex
ual behavior.’ In primates, small
dietary exposures to dioxin are associ
ated with an increased risk and severity
of endometriosis.4 A study in humans
also shows higher levels of dioxin in
women with endometriosis than in a
control population?
Dioxin is particularly toxic to the
developing immune system. Animal
tests show that nanograms per kilo
gram doses given 1-4 times during
pregnancy cause permanent alterations
in the immune system of offspring?
Human studies also show an increased
susceptibility to infection and changes
in immune system parameters as a
result of in utero exposure to ambient
environmental levels of dioxin and
dioxin-like compounds.7, s Low levels
of exposure during pregnancy also alter
thyroid hormone levels in mothers and
offspring, perhaps explaining neurologi
cal effects, including learning disabili
ties, that are seen in carefully
conducted primate studies?
How are we exposed?
The US EPA estimates that over 90%
of our exposure is through food, with
major sources including beef, dairy
products, fish, pork, and breast milk.
What is the Level of
exposure in the general
population?
The general population, through ordi
nary dietary exposures, carries a cur
rent body burden of dioxin that is near
or above the levels that cause adverse
effects in animal tests. Through food
alone, Americans are getting 22 times
the maximum daily dioxin exposure
considered by the US EPA to be with
out adverse effects.
Breast milk contamination is such that
the nursing infant, during vulnerable
periods of development, is exposed to
dietary levels of dioxin 35 to 65 times the
amount considered safe. Nonetheless,
breast feeding remains far superior to
formula feeding for a variety of rea
sons, and reducing breast feeding is
not an appropriate public health
response.
What are the
sources of dioxins?
Dioxins are unintentionally formed
during a variety of industrial processes.
Dioxin-like compounds can be gener
ated and released to rhe environment
from various combustion processes
when chlorine donor compounds are
present. Chlorine donor compounds
can include polyvinyl chloride (PVC)
plastic and other chlorinated com
pounds. Dioxin compounds can also
be formed during the manufacture of
chlorine and chlorine-containing com
pounds including the monomers which
comprise PVC, chlorinated solvents
and pesticides. Dioxins can also be
formed during the bleaching of paper
with chlorine, and in other industrial
and combustion processes that include
rhe presence of chlorine.
The primary source of dioxins from the
health care sector is waste incineration.
Chlorine-containing products burned
in incinerators, including medical
devices and products, provide the chlo
rine necessary for dioxin formation.
Prior to rhe implementation of new
rules (which will reduce the health
care sector’s contribution to total diox
in loading) the EPA identified munici
pal and medical waste incinerators as
two of the leading sources of dioxin
emissions to air in the US.
Once dioxin is emitted into the air
from incinerators and other sources,
rain, snow and dust can carry it to the
surface of the earth, where it can enter
the food chain.
What is the evidence
that the manufacture of
PVC feedstocks is linked
to dioxin formation?
The draft dioxin reassessment recently
released by the US Environmental
Protection Agency (EPA) reviews the
contribution of PVC manufacturing to
dioxin emissions.10 According to cal
culations of the Vinyl Institute (an
industry' trade association), reviewed
and given a medium confidence rating
by the EPA,11 the production of PVC
and its feedstocks result in air releases
of 11.2-31.0grams toxic equivalency
(TEQ)1- dioxins and furans per year.
These levels may understate the con
tribution of dioxin from the manufac
ture of PVC throughout its lifecycle.
Under what conditions
can the combustion of PVC
result in dioxin formation?
The draft EPA dioxin reassessment
also reviews the contribution of waste
incineration to dioxin emissions. The
report summarizes a large body of liter
ature that finds carbon and catalysts
must be present in an incinerator in
order for dioxins to form.” PVC is
usually the largest chlorine source in
municipal and medical waste incinera
tors. The relationship between chlo
rine inputs into an incinerator and
dioxin formation, however, depends
upon combustion conditions.
For uncontrolled combustion, such as
open burning of household waste,
landfill fires, or building fires, a direct
association between chlorine content
of the combusted material and dioxin
formation has been established. For
example, a study of the open burning
of household waste showed that waste
containing larger amounts of PVC
(4.5% vs. 0.2%) produced substantially
larger amounts of dioxins in air emis
sions (269 vs. 44.3 microgram/kg waste
burned) and ash (7,356 vs. 489 micro
gram/kg waste burned).'4
In modern commercial waste incinera
tors, the rate at which dioxins are
formed and released depends upon
chlorine inputs, incinerator design,
operating conditions, the presence of
catalysts, and pollution control equip
ment. While the EPA concludes, based
on studies of modern waste incinera
tors, that the largest determinants of
dioxin formation are operating condi
tions (including overall combustion
efficiency, post-combustion flue gas
temperatures, and residence times —
and the presence of iron or copper cat
alysts) rather than chlorine content
alone, there is little doubt that chlorine
content ol the waste feed is critical.
Several laboratory and incinerator
(
pilot studies have found a direct rela
tionship between chlorine loading and
dioxin emissions.IS In addition, the
EPA’s conclusion appears to rest large
ly on an analysis of incinerator emis
sions data by Rigo, et al. (1995), which
has serious methodological flaws.16 It
is also important to note that the EPA
conclusion refers only to stack gas
emissions, which are a relatively small
fraction of total dioxins released from
incinerators, and does not consider
releases in fly ash, bottom ash, and
water discharges.
For any given waste incinerator,
according to the EPA, conditions may
exist in which changes in chlorine
content of waste feed will correlate
highly with dioxin and furan emis
sions. These conditions may prevail
during start-up or shut-down, changes
in waste feed rate, or operational
upsets. Although modern commercial
waste incinerators are designed and
intended to be operated to minimize
release of dioxins and other hazardous
air pollutants, they are, nevertheless, a
significant source of dioxin releases.
What is Health Care
Without Harm's position
on dioxin, PVC, and med(fccal waste incineration?
Available data reveal a complex rela
tionship among chlorine feed, design
and operating conditions, and dioxin
emissions. It is certain that chlorine
sources are necessary for dioxin emis
sions, PVC products are the largest
chlorine source, and incinerators with
pollution control equipment are signif
icant sources of dioxin releases in
stack gases, fly ash, bottom ash, and
water discharges. Moreover, even
modern, well-designed incinerators do
not consistently operate at optimal
combustion conditions.
For these reasons, along with concern
about other hazardous pollutants emit
ted from waste incinerators — includ
ing mercury, particulates, sulfur and
nitrous oxides, and hydrochloric acid
— Health Care Without Harm has
taken the pollution prevention posi
tion that PVC use should be mini
mized and ultimately eliminated,
alternatives used when available with
out compromising patient safety or
care, and all unnecessary waste incin
eration should be avoided.
Notes
1.
2.
Brown NM, Manzolillo PA, Zhang JX, et al.
Prenatal TCDD and predisposition to mammary cancer in the rat. Carcinogenesis
19(9): 1623-1629, 1998.
Steenland k, Piacitelli L, Deddens J, et al.
Cancer, heart disease, and diabetes in work
ers exposed to 2,3,7,8-tetrachlorodibenzo-pdioxin J Natl Cancer Inst 91(9);779-786
1999.
3.
Mably TA, Moore RW, Peterson RE. In
utero anil lactational exposure of male rats
to 2,3,7,8-tetrachlorodibenzo-p-dioxin. 1.
Effects on androgenic status. Toxicol Appl
Pharmacol 114:97-107, 1992; and Schantz
SL, Bowman RE Learning m monkeys
exposed perinatally to 2,3,7,8-tetrachlorodibcnzo-p-dioxin (TCDD).
Neurotoxicol Teratol 11 (1) • 13-19, 1989.
4.
Rier SE, Martin DC, Bowman RE, et al.
Endometriosis in Rhesus monkeys (Macaca
mulatta) following chronic exposure to
2,3,7,8 -tetrachlorodibenzo-p-dioxm. Fund
Appl Toxicol 21:433-441, 1993.
5.
Mayani A, Barcl S, Soback S, Almagor M.
Dioxin concentrations in women with
endometriosis. Human Reprod 12(2):373375, 1997.
6.
Birnbaum LS. Workshop on perinatal expo
sure to dioxin-like compounds. V
Immunologic effects. Environ Health
Perspect 103(suppl 2)157-160, 1995.
7.
Weisglas-Kuperus N, Koopman-Esseboom C,
et al. Immunologic effects of background
prenatal and postnatal exposure to dioxins
and polychlorinated biphenyls in Dutch
infants. Pediatr Res 38:404-410, 1995.
8.
Weisglas-Kuperus N, Patandin S, Berbers G,
et al Immunologic effects of background
exposure to polychlorinated biphenyls and
dioxins in Dutch preschool children.
Environ Health Perspect 108(12); 12031207, 2000.
9.
Koopman-Esseboom C, Morse DC,
Weisglas-Kuperus N, et al. Effects of dioxins
and polychlorinated biphenyls on thyroid
status of pregnant women and their infants.
Pediatr Res 36(4):468-473, 1994.
10. See US EPA, Report #: EPA/600/P00/001 Ab, March 2000
11 The EPA developed a three-part confidence
rating scheme: “high” means the estimate is
derived from a comprehensive survey,
“medium’’ is based on estimates of average
activity and number of facilities or a limited
survey; and "low” is based on data judged
possibly non-representative
12. Since the toxicity of the various congeners
of dioxins anil furans vanes, the toxicity of a
given mixture of congeners is usually
expressed as TEQs, where the most toxic
form is assigned a value of one and the rela
tive contribution of others is calculated
accordingly.
13. Dioxins/furans form most readily in com
mercial incinerators as the combustion gases
reach cooler temperatures, primarily in the
range 2OO-45O°C.
x
14. Lemieux PM. Evaluation of emissions from
rhe open burning of household waste in bar
rels. USEPA. EPA/6OO/SR-97/134, 1998.
15. For example, see: Bruce, et al, The role of
gas phase C12 in the formation of
PCDD/PCDF during waste combustion,
Waste Management, 11: 97-102, 1991;
Kanters, et al, Chlorine input and
chlorophenol emission in the lab-scale com
bustion of municipal solid waste,
Environmental Science and Technology, 30:
2121-2126, 1996; and Wagner and Green,
Correlation of chlorinated organic com
pound emissions from incineration with
chlorinated organic input, Chemosphere,
26: 2039-2054, 1993.
16. In 1995, the Vinyl Institute commissioned a
report, prepared for the American Society
of Mechanical Engineers, that purported to
examine the relationship between PVC in
incinerator waste feed and dioxin emissions
(Rigo HG, Chandler JA, Lanier WS, The
relationship between chlorine in waste
streams and dioxin emissions from combus
tors, The American Society' of Mechanical
Engineers, 1995). After examining data
from dozens of burns in a number of munic
ipal and medical waste incinerators, rhe
report concludes that there is no statistical
ly significant relationship between fuel chlo
rine content and dioxin emissions. The
analysis, however, is flawed in a number of
significant ways. First, there was no attempt
to control for differences in incinerator
design or operating conditions so that the
question of interest could be addressed
independent of other variables. Second,
the authors used data collected for regulato
ry compliance purposes and not intended to
examine the relationship between chlorine
input and dioxin output. Without actuallyknowing the PVC content of the waste
feed, they were forced to use hydrochloric
acid emissions as a surrogate for chlorine
loading Hydrochloric acid emissions can
be used to approximate chlorine loading but
do not provide precise estimates. Moreover,
in the tested incinerators, dioxin concentra
tions were sampled at various points in the
exhaust stream - from boiler outlet to fur
ther downstream - predictably a source of
variability, since dioxin can be formed at
various points in the exhaust, depending on
temperature and fly ash composition. This
sampling strategy provides a poor estimate
of total dioxin emissions to the air and ash.
In summary', this analysis relies on data that
are poorly suited to answer the question of
interest. A more complete referenced dis
cussion of the connection between PVC
incineration and dioxin formation may be
found in: Thornton J., Pandora’s Poison:
Chlorine, Health, and a New
Environmental Strategy (Chapter 7), MIT
Press: Cambridge MA, 2000.
r~>
>
m
on
What's
Wrong With
Incineration?
Health Care Without Harm has several
concerns regarding the burning of
waste generated by health care (both
solid waste and regulated medical
waste). Incineration produces both
toxic air emissions and toxic ash
residue.1 The air emissions affect the
local environment, and in many cases,
may affect communities hundreds or
thousands of miles away. The ash
residue is sent to landfills for disposal,
where the pollutants have the potential to leach into ground water. (It must
be noted that waste treated by other
methods and then landfilled will also
produce leachate.)
In addition to releasing the pollutants
contained in the waste stream to the
air and into the ash, burning medical
waste actually creates new toxic com
pounds, such as dioxins. Medical waste
incineration has been identified by the
U.S. Environmental Protection Agency
as the third largest known source of
dioxin air emissions,2 and as the con
tributor of about 10 percent of the
mercury emissions to the environment
from human activities?
Many, if not most, on-site medical
waste incinerators burn not only infec
tious waste, but also readily recyclable
items such as office paper and card
board. This destroys resources and pre
vents cost savings that could be
recouped through recycling. Medical
waste incineration’s identification as a
primary source of some very toxic pol
lutants stands in direct contradiction
to physicians’ oaths to “do no harm.”
Dioxin
o
o
cc
Dioxin belongs to a family of 419
chemicals with related properties and
toxicity, but the term “dioxin" is often
used to refer to the 29 that have simi
lar toxicity. Dioxin is one of the most
toxic chemicals known to humankind.
While exposure of the general popula
tion occurs through the ingestion of
many common foods, children exposed
in utero during critical periods of devel
opment appear to be the most sensitive
and vulnerable to the effects of dioxin?
Dioxin exposure has been linked to
disrupted sexual development, birth
defects and damage to the immune sys
tem. Dioxin has been associated with
IQ deficits, hyperactive behavior and
developmental delays?-6
The International Agency for Research
on Cancer (IARC), an arm of the
World Health Organization, acknowl
edged dioxin’s cancer-causing potential
when they classified it as a known
human carcinogen? The U.S.
Environmental Protection Agency
(EPA) has determined that most
Americans are exposed to dioxin
through ingestion of common foods,
mostly meat and dairy products. Dairy
cows and beef cattle absorb dioxin by
eating contaminated feed crops. The
crops become contaminated by air
borne dioxins that settle onto soil and
plants. Dioxins enter the air from
thousands of sources including inciner
ators that burn medical, municipal and
hazardous waste?
Mercury
Mercury is a potent neurotoxin, which
means it attacks the body’s central
nervous system; it can also harm the
brain, kidneys and lungs. It can cross
the blood-brain barrier as well as the
placenta. Mercury poisoning can cause
slurred speech, impaired hearing,
peripheral vision and walking, muscle
weakness, mood swings, memory loss
and mental disturbances. The risks of
damage to the nervous systems of
developing fetuses and young children
are primary reasons for fish-consump
tion advisories, aimed at discouraging
pregnant women, women of child-hear
ing age, and young children from eat
ing too much fish. Studies done on
women who ate methylmercurycontaminated fish or grain showed
that even when the mothers showed
few effects of exposure, their infants
demonstrated nervous-system damage.
If mercury-containing items are put
into a "red bag” for infectious waste
and sent to an incinerator, mercury' will
504- 508; Weisglas-Kupcrus N, Sas TCJ,
Koopman-Esseboom C, et al. 1995
“Immunologic effects of background prena
tal and postnatal exposure to dioxins and
polychlorinated biphenyls in Dutch infants.”
Pediatr Res 38. 404-410; Huisman M,
Koopman-Esseboom C, Fidler V, et al. 1995.
“Perinatal exposure to polychlorinated
biphenyls and dioxins and its effect on
neonatal neurological development." Early
Human Development 41: 111-127.
contaminate the air. (This can happen
with non-incinerauon technologies as
well. If mercury goes into treatment
equipment, it will come out.) Airborne
mercury then enters a global distribu
tion cycle in the environment, contam
inating fish and wildlife.
Other Hazardous
Pollutants
Many other hazardous pollutants have
been identified in the emissions from
medical waste incinerators: arsenic,
ammonia, benzene, bromodichlor
omethane, cadmium, carbon tetrachlo
ride, chromium, chlorodibromomethane, chloroform, cumene, 1,2dibromoethane, dichloromethane,
dichloroethane, ethyl benzene, lead,
mesitylene, nickel, particulate matter,
naphthalene, tetrachloroethane,
toluene, trichloroethane, 1,1,1trichloroethane, trichloroethylene,
trichloromethane, vinyl chloride, and
xylenes." Analysis of emissions of other
treatment methods is necessary to
determine if these emissions occur in
the absence of combustion.
5
“Workshopfs] on Perinatal Exposure to
Dioxin-like Compounds I-VI.
Summar(ies),” Environmental Health
Perspectives Supplements, Vol. 103,
Supplement 2, March 1995.
6.
Health Assessment Document For 2,3,7,8Tetrachlorodiben<o-P-Dioxin (TCDD) Anil
Related Compounds, Vol. 1 of III, and Vol. II
of III, USEPA, Office of Research and
Development, EPA/600/ BP-92/00 lb and
EPA/600/BP-92/001c, external review draft,
and Devito, M J and Birnbaum, L S. (1994)
“Toxicology of dioxins and related chemi
cals " In Dioxins And Health, Arnold
Schecter, ed., NY: Plenum Press, 139-62, as
cited in Dying From Dioxin. A Citizen's Guide
To Reclaiming Our Health And Rebuilding
Democracy, Gibbs, L M and the Citizens
Clearinghouse for Hazardous Waste, Boston:
South End Press, 1994, pp. 138-139.
7.
“1ARC Evaluates Carcinogenic Risk
Associated with Dioxins," International
Agency for Research on Cancer press
release, February 14. 1997.
8.
Estimating Exposure To Dioxin-Like
Compounds, Volume 1: Executive Summary’,
USEPA, Office of Research and
Development, EPA/600/6-88/005Ca. June
1994 review draft, p. 36.
9.
Draft Technical Support Document To Proposed
Dioxins And Cadmium Control Measure For
Medical Waste Incinerators, California Air
Resources Board, 1990, pg.51, as cited in
“Medical Incinerators Emit Dangerous
Metals And Dioxin, New Study Says,”
Rachel’s Environment &. Health Weekly
#179, May 2, 1990.
References
1
“Issues in Medical .Waste Management
Background Paper,” Office of Technology
Assessment, Congress of the United States,
OTA-BP-O-49, October, 1988.
2.
Inventory of Sources of Dioxin in the United
States (EPA/600/ P-98/002 Aa), National
Center for Environmental Assessment,
USEPA, April 1998, p. 2-13.
3.
Mercury Study Report to Congress, Volume I:
Executive Summary, USEPA Office of Air,
December 1997, pp 3-6.
4.
Pluim, HJ, Koope, JG, Olie, K., et al. 1994.
“Clinical laboratory manifestations of expo
sure to background levels of dioxins in the
perinatal period.” Act Paediatr 83:583-587,
Koopman-Esseboom C, Morse DC, WeisglasKuperus N, et al. 1994. “Effects of dioxins
and polychlorinated biphenyls on thyroid
hormone status of pregnant women and
their infants.” Pediatr Res .36: 468-473; Pluim
HJ, de Vijlder JJM, Olie, K, et al. 1993.
“Effects of pre- and postnatal exposure to
chlorinated dioxins and furans on human
neonatal thyroid hormone concentrations.”
Environmental Health Perspectives 101 -
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green:
Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit. or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm org.
Il.v IX F ccnitkanon mark mJ term .ire cb.v sA pn^vrty at the Chlorine Free
ProJucti Association and are only used by authorized and ccttil.ed ux-r»
Order form for
Alternative
Medical Waste
Treatment
Technologies
Table of Contents
Preface
Executive Summary
1.
Introduction: Why Non-incineration Technologies?
2.
Strategic Framework for Non-incineration Technologies: The Broader Context
3.
Understanding the Waste Stream: A Necessary First Step
A Resource for Hospital
Administrators, Facility
Managers, Health Care
Professionals, Environmental
Advocates, and Community
Stakeholders
4.
Non-incineration Technologies: General Categories and Processes
5.
Low-Heat Thermal Technologies: Autoclaves, Microwaves, and Other SteamBased Systems
6.
Low-Heat Thermal Technologies: Dry Heat Systems
7.
Medium- and High-Heat Thermal Technologies: Depolymerization, Pyrolysis,
and Other Systems
8.
Chemical-Based Technologies: Chlorine and Non-Chlorine Based Systems
Published August 2001
9.
Irradiation, Biological, and Other Technologies: E-Beam, Biological, and
Sharps Treatment Systems
10. Factors To Consider in Selecting a Non-incineration Technology
11. Economics of Treatment Technologies: Comparing Treatment Options
12. References and Recommended Readings
Appendices
This publication was printed in August 2001 and can be ordered by completing
the form below and sending it by postal mail or via fax to:
Jolie Patterson, Health Care Without Harm
1755 S Street, NW, Suite 6B, Washington, DC 20009
Phone: 202.234.0091 Fax: 202.234.9121
You may also email your request to info@hcwh.org. Please supply the following
information.
Donations to cover the cost of printing and postage are suggested. Please send your
check or money order for $15 made payable to “Health Care Without Harm.”
Name:
O rga niza t ion:
Ad d ress:________
City, State/Province, Zip Code:
Phone: ()Fax:()
E-mail: _ _____________________________________________________________
Reducing
Polyvinyl
Chloride (PVC)
Use in
Hospitals
There are many ways in which hospi
tals can take immediate action to
reduce PVC use. The process will
involve:
n
gathering data through audits and
letters to vendors;
n
identifying alternatives;
n
developing and implementing a
PVC reduction plan; and
n
establishing a PVC reduction policy.
Begin by identifying
products that contain
PVC and determining
appropriate alternatives
Reducing PVC requires knowing which
products contain PVC and the avail
ability of alternatives. PVC products
range from critical health care devices,
such as disposable intravenous (IV)
bags and tubing, to bedpans and note
book binders, as well as basic construc
tion materials and furnishings, such as
water pipes and wall coverings. If you
take the time to identify products your
hospital purchases and the materials
that they are made of, it will facilitate
the process of reducing PVC use over
time. For example, Catholic
Healthcare West, a large nonprofit hos
pital system, requires its group purchas
ing organization (GPO) to identify
products that contain PVC.
To start a list of PVC products in your
hospital see Table 1 and the
Sustainable Hospitals Project website,
www.sustainablehospitals.org.
What should be included
in a PVC reduction plan?
Reduction priorities should be based
on the potential for patient exposure to
DEHR potential for the PVC product
to be incinerated upon disposal, vol
ume of PVC use, and availability of
substitute products.
Taking into consideration these con
cerns, it is wise to establish an organi
zation-wide PVC reduction plan that
includes the following priorities:
a. First, target disposable PVC health
care products, especially within
neonatal intensive care units
(NICUs), maternity departments,
and pediatrics.
b. Second, phase out the purchase of
PVC office supplies.
c. Third, purchase PVC-free furnish
ings, furniture products, and con
struction products when purchasing
new furniture, renovating existing
departments, or constructing new
wings or buildings; and
d. Fourth, when buying new durable
medical products, specify those that
are PVC-free.
Disposable PVC health care products
should be the first priority because of
the potential for significant patient
exposure to DEHP and because they
may be incinerated at the end of their
useful life. DEHP exposure is critical
to consider, especially for fetuses, new
borns, and toddlers who may be
exposed to levels of DEHP near or at
those that cause harm in relevant ani
mal models. Since DEFIP is a repro
ductive and developmental toxicant,
DEFIP use in NICUs, maternity
departments and pediatrics is of partic
ular concern. For maternity depart
ments, NICUs, and pediatrics, health
care providers may decide that elimi
nating DEHP exposures in their partic
ularly vulnerable patients justifies the
higher cost for some of the alterna
tives.
Office supplies are another priority for
elimination because they may be incin
erated upon disposal, cost-competitive
alternatives are widely available, and
hospitals usually can replace them eas
ily under existing contracts.
PVC-containing furnishings, furni
ture products, and construction prod
ucts should be eliminated from new
purchases, building renovations, and
new building construction. For most of
these products, cost-competitive, PVCfree alternatives are widely available.4
Durable medical products pose the
greatest challenge to reduction due to
the lack of knowledge of their PVC
content and availability of PVC-free
devices. The primary use for PVC in
durable medical products is as the
housing — the rigid, outer plastic cov
ering — for testing and diagnostic
equipment. Since durable medical
products have a longer use life than
disposable medical products (such as
IV bags) and result in little DEHP
exposure, they are a secondary target
for reduction. A first step in reducing
PVC use in these applications would
be to require vendors to disclose the
PVC content in their equipment.
How do PVC-free
and DEHP-free
alternatives differ?
DEHP-containing PVC: Because
PVC is a rigid plastic by nature, manu
facturers add DEHP to make PVC
flexible. DEHP does not chemically
bind to PVC. DEHP may therefore
leach from plasticized PVC when a
medical device comes into contact
with fluids, lipids, and/or heat. DEHP
is a reproductive and developmental
toxicant in laboratory animal testing.
Other toxicity concerns are unre
solved. [See, Health Care Without
Harm’s Fact Sheet, “DEHP Exposure
During the Medical Care of Infants: A
Cause for Concern.’’]
PVC-free: Non-PVC plastics used in
medical devices include silicone, poly
ethylene, and polypropylene. Mostflexible, PVC-free medical devices do
not contribute chlorine to waste incin
erators and are, therefore, less likely to
contribute to dioxin formation when
waste is burned. In addition, PVC-free
products do not contain plasticizers,
and potential risks from plasticizer
leaching are avoided.1
DEHP-free: DEHP-free PVC medical
devices contain alternative softening
agents (plasticizers), such as citrates
and trimellitates, which have been
substituted for DEHP Both may leach
from PVC, although at different rates,
depending on the nature of the solu
tion in the bag. Citrates are less haz
ardous than DEHR as indicated by
their use as a food additive. Much less
is known about rhe safety/hazards of
the trimellitates, though some research
indicates that trimellitates leach less
than DEHP’-’ While purchasing
DEHP-free PVC products is an option
for reducing DEHP exposure, it should
only be considered an interim solution
because it does not address the lifecy
cle impacts of PVC.
Which disposable
PVC health products
contain PVC and what
are the alternatives?
Disposable PVC health care products
fall into five broad categories: bags,
tubes, gloves, trays,5 and catheters.
Bags (42.5%), tubes (43.0%), and
gloves (12.5%) account for 98% of dis
posable PVC healthcare products.6
PVC bags package IV products, enter
al feeding formulas, and blood products
(including packed red blood cells, fresh
frozen plasma, and platelet rich plas
ma). PVC bags are also used to collect
bodily fluids. DEHP-containing PVC
medical bags first became a matter of
concern in the 1970s because of DEHP
exposures from the use of blood and
total parenteral nutrition (TPN) bags.
Alternatives to PVC bags: PVC-free
bags are on the U.S. market for pack
aging IV products, platelet rich plas
ma, fresh frozen plasma, enteral formu
la, and TPN. The PVC-free bags are
both cost- and technically-competitive
with the PVC bags.
For packed red blood cell bags, howev
er, there is only a DEHP-free alterna
tive. An unintended consequence of
DEHP leaching from PVC bags is that
it acts as a preservative of red blood
cells by extending the shelf-life of
stored red blood cells. The Food and
Drug Administration does not regulate
DEHP as an additive to red blood
cells. The alternative plasticizer used
in red blood cell bags is a citrate.
Citrates, in fact, have a long history of
use as a blood preservative. The shelf
life of blood in citrate-plasticized bags
is similar to that of DEHP-plasticized
bags. A DEHP-free bag is on the mar
ket at a slightly higher cost than the
DEHP-containing PVC bag.
PVC tubing conveys liquids — such
as IV solutions and nutritional formu
las — and respiratory gases to
patients. PVC tubing and catheters are
actually poor technical performers in
medical treatments that involve con
tact with human tissue longer than
approximately three to seven days.
The leaching of DEHP not only expos
es patients to the plasticizer, but also
causes the product to become brittle
and subject to cracking. For these rea
sons, products like umbilical vessel
0)
catheters and gastrostomy tubes are no
longer manufactured from PVC.
Recent research suggests that signifi
cant levels of DEHP may leach out of
nasogastric tubes within 24 hours. A
Swedish study of PVC nasogastric
tubes used for 24 hours “showed that
the section of rhe tube which had been
inside the infant’s stomach contained
only half as much plasticiser as the rest
of the tube. ...Since this discovery,
the [Swedish County] council’s med
ical board decided to substitute
polyurethane tubes for the PVC
ones.”7
Respiratory Therapy Products
n aerosol and oxygen masks,
tents, and tubing
° endotracheal and tracheostomy
tubes
a humidifiers, sterile water bags
and tubing
° nasal cannulas and catheters
n resuscitator bags
° suction catheters
Collection of Bodily Fluids
n dialysis, peritoneal: drainage bags
n urinary’ collection bags, urologi
cal catheters, and irrigation sets
° wound drainage systems: bags
and tubes
Enteral Feeding Products
D enteral feeding sets (bags and
tubing)
n nasogastric tubes
a tubing for breast pumps
n notebook binders
n plastic dividers in patient charts
Gloves, Examination
Durable Medical Products
Intravenous (IV) Therapy Products
a catheters
a solution bags
n tubing
Packaging, Medical Products
d film wrap
□ thermoformed trays for admis
sion and diagnostic kits, and
medical devices
Patient Products
□ bed pa ns
□ cold and heat packs and heat
ing pads
■ inflatable splints and injury' sup
port packs
s patient ID cards and bracelets
■ sequential compression devices
□ testing and diagnostic equip
ment, including instrument
housings
Furniture Products and
Furnishings
° bed casters, rails, and wheels
a floor coverings
a furniture upholstery'
■ inflatable mattresses and pads
u mattress covers
a pillowcase covers
a shower curtains
a thermal blankets
° wallpaper
n window blinds and shades
Construction Products
° doors
■ electrical wire sheathing
o pipes: water and vent
■ roofing membranes
■ windows
H O S P IT A
Kidney (Renal Disease)
Therapy Products
u hemodialysis: blood lines (tub
ing) and catheters
□ peritoneal dialysis: dialysate
containers (bags)
and fill and drain lines (tubing)
Office Supplies
IN
PVC-free construction and furnishing
products are widely available and are
often cost-competitive. For example,
PVC-free mattress covers and shower
curtains can be purchased and are
cost-competitive with the PVC prod
ucts. During renovations and new
building construction, hospitals should
Blood Products and Transfusions
n apheresis circuits
n blood bags and tubing
D extracorporeal membrane oxy
genation circuits
USE
Are PVC-free construction
and furnishing products
available?
Disposable Health
Care Products (continued)
(P V C )
Alternatives to PVC gloves: Latex is
the dominant material used in the
manufacture of examination gloves.
However, concerns with latex allergies
have led hospitals and manufacturers
to consider gloves made of different
materials. For example, when Kaiser
Permanente decided to phase-out the
use of latex gloves it searched for
PVC-free gloves, ultimately settling on
gloves made of nitrile. While these
are more expensive than latex and
PVC gloves, Kaiser received a costcompetitive bid due to the size of its
^contract. Reflecting growing demand,
diversity’ of latex-free and PVC-free
gloves is on the market today,
although costs are slightly higher.9
Disposable Health
Care Products
C H L O R ID E
PVC gloves: PVC is used primarily in
Jfche manufacture of examination gloves
imd has little market share in the sur
gical glove market.
Table l.? Polyvinyl Chloride (PVC) Products in Hospitals
L Y V IN Y L
Alternatives to PVC tubing: PVCfree or DEHP-free tubing is on the US
market for most medical applications.
Silicone, polyethylene, and
polyurethane are three alternative
polymers frequently used in tubing
applications. In most applications, at
least one of these polymers can com
pete with PVC in terms of technical
performance. In terms of economic
performance, PVC-free tubing general
ly costs more than PVC tubing. In the
next few years, however, plastics indus
try’ analysts expect metallocene poly
olefins (polyethylene and polypropy
lene are polyolefins) to become costcompetitive with flexible PVC medical
products.'
specify PVC-free products. Including
home and commercial buildings, con
struction products, furnishings, and
furniture products account for approxi
mately 75% of all PVC end uses.
Why establish an
organization-wide PVC
reduction policy?
An organization-wide PVC reduction
policy is an important step toward
eliminating PVC products from hospi
tals because it reflects senior manage
ment’s support for action, signals staff
to take the issue seriously, and illus
trates to vendors the need to market
PVC-free products. Educational pro
grams - workshops, grand rounds, and
conferences - can raise staff and man
agement’s awareness of the lifecycle
hazards of PVC and rhe toxicity of
DEHP The time investment in plan
ning and education internally can
result in broader PVC reduction poli
cies. For example, Tenet Healthcare
and Universal Health Services entered
into memoranda of understanding
between management and shareholders
on reducing PVC use throughout their
hospital systems after learning about
the hazards of PVC.
Tenet Healthcare agreed to: “investi
gate the availability and utility of PVCfree and phthalate-free disposable
medical products available in the mar
ketplace;” “seek information on a regu
lar basis from its suppliers of disposable
medical products concerning whether
their products are PVC-free and
phthalate-free;” and “request its suppli
ers of disposable medical products to
aid in the development of and further
advancements in PVC-free and phthalate-free disposable medical products.”
Notes
1.
A few PVC-free products do contain chlo
rinc, including neoprene gloves, which are
manufactured from polychloroprene.
2.
Chnstensson A, Ljunggren L, NilssonThorcll C, Arge B, Diehl U, Hagstam KE,
Lundberg M. In vivo comparative evaluation
of hemodialysis tubing plasticized with
DEHP and TEHTM. Ini J Artif Organs
14(7):4O7-1O, 1991.
3.
Quinn MA, Clync JH, Wolf MM,
Cruickshank D, Cooper IA, McGrath KM,
Morris J. Storage of platelet concentrates—
an in vitro study of four types of plastic
packs. Pathology 18(3):331-5, 1986.
4-
Currently wire and cable coated with PVC is
the most difficult of these products to
replace.
5.
Trays arc used to package surgical instru
ments, kits for surgical procedures, medical
diagnostic kits, and admission kits.
6.
Schlechtcr, M. Plastics for Medical Devices:
What's Ahead? Norwalk, CT Business
Communications Company, Inc., 1996.
7.
The Federation of Swedish County
Councils, PVC in the Swedish Healthcare
System, Stockholm, 2000.
8.
“The PVC markets that are specifically tar
geted tor replacement [by metallocene poly
olefins! include flexible medical uses, pack
aging film, wire and cable insulation, trans
portation, flooring and geomembranes”
(Aida M. Jebens, 1997, Chemical Economics
Handbook: Polyvinyl Chloride (PVC)
Resins, Palo Alto: SRI International, p.
580.1882B).
9.
For a list of products see.
www.sustainablehospitals.org.
HealthrCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: /I Resource Kit for Pollution
Prevention in Health Care, For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at vAvw.noharm.org.
Flic ICF cettilicanon mark and term are the sole property of the Chlorine Free
1 roducti Aviation anti are only uxd by authored and certified tncr>
Alternatives*
to Polyvinyl
Chloride (PVC)
and
Di-2-Ethylhexyl
Phthalate
(DEHP)
Medical
Devices
Products detailed in this
publication include:
Ambulatory Products
Gloves, Examination
Intravenous (IV) products:
■ administration sets
H bags
■ infusion tubes
Bedding Products
Patient ID Bracelets
Blood bags:
Q fresh frozen plasma
B packed red blood cells
B platelets
B platelet rich plasma
Respiratory Therapy Products
H endotracheal tubes
■ masks, aerosol and oxygen
■ oxygen hood
■ tracheostomy tubes
Body Bags
Sequential Compression Devices
Central line catheters and PICC lines
B introcan safety catheters
a midline catheters
a percutaneous catheter introducers
n peripherally-inserted central
catheters (PICC)
Total parental nutrition
■ bags
B catheters
0 tubing
Umbilical vessel catheters
Dialysis, peritoneal
■ rigid dialysate containers
■ peritoneal catheters
Enteral feeding sets
° bags and tubing
H extension sets
Enteral feeding nasogastric tubes
■ PEG tubes
■ gastrostomy tubes
■ nasoenteric tubes
■ nasogastic tubes
■ nasojejunal tubes
■ pediatric clear straight catheters
Urinary drainage catheters
B Foley catheters
■ urethral catheters for pediatrics
B urinary catheters
Wound Drains and Drainage Systems
■ drains
■ nephrostomv catheters
■ surgical and wound drains
■ thoracic catheters
Office Supplies
Shower Curtains
Epidural vessel catheters
* Health Care Without Harm does not endorse any of these products, has not tested them
for safety or efficacy, and does not take responsibility for the accuracy of the information
or product performance. Listing here is based solely on information provided by the man
ufacturer. Non-PVC products may contain much smaller amounts of DEHR Flexible
PVC-free products still must be tested to ascertain whether they are in fact DEHP-free.
Products that contain latex and chlorine are excluded from this table: latex products
because of concerns over latex allergies and chlorine containing products because of con
cerns over lifecycle hazards. Exceptions are made for the few PVC products for which few
or no non-PVC products are available. In those cases non-DEHP products are identified.
This table is a work-in-progress.
Sources: Sustainable Hospitals Project, 2000, “Alternative Products." see http://sustainablehospitals.org (Lowell: Sustainable Hospitals Project, UMass Lowell); and Tickner, Joel,
et al. 1999, The Use of Di-2-Ethylhexyl Phthalate in PVC Medical Devices: Exposure,
Toxicity, and Alternatives (Lowell: Lowell Center for Sustainable Production, UMass
Lowell); and all information was verified through telephone contacts with manufacturer
representatives or review of manufacturer website information.
ALTERNATIVES
TO
PVC
AND
DE HP
MEDICAL
DEVICES
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 1 of 4)
Products
Manufacturer
Telephone
Webpage
Material
Comments
Ambulatory Products
Many manufacturers including
Merry Walker Corp.
815-678-3388
www.merrywalker.com
Steel
Product: Merry Walker
Bedding Products
Precision Dynamics Corp.
800-847-0670
www.pdcorp.com
Polyethylene
Disposable mattress and pillow
covers, draw sheets
Blood Bags
Baxter Healthcare, Fenwal Division
800-766-1077
www.baxter.com
Polyolefin
Bags for platelets, platelet rich plas
ma and fresh frozen plasma
Bags for packed red blood cells
Non-DEHP PVC
Body Bags
LASAN Plastics, Inc.
207-693-4817
www.lasan.com
Polyethylene/polypropylene blend
Central Line Catheters
and PICC Lines
B. Braun
800-227-2862
www.bbraunusa.com
Polyurethane or Teflon
Teflon or polyurethane
Percutaneous catheter introducers
Central venous catheter, introcan
safety catheter
Becton Dickinson
201-847-6800
www.bd.com
Silicone or polyurethane
Peripherally-inserted central
catheter, midline catheter
Klein-Baker Medical
210-696-4061
www.neocare.com
Silicone
Peripherally-inserted central
catheter (neonates)
Utah Medical Products, Inc.
800-533-4984
www.utahmed.com
Silicone
Peripherally-inserted central
catheter (neonates)
Vygon
800-544-4907
www.vygonusa.com
Polyurethane or Silicone
Peripherally-inserted catheter
(adults and neonates)
Midline catheters (pediatrics or
adults)
Polyurethane
Dialysis, Peritoneal
Enteral Feeding Sets
B. Braun
800-621-0445
www.bbraunusa.com
Polypropylene/polyethylene comonomer
Rigid peritoneal dialysate container
Degania Silicone
401-658-0130
www.deganiasilicone.com
Silicone
Peritoneal catheter
Children's Medical Ventures
800-377-3449
www.childmed.com
Non-DEHP PVC
Enteral set
CORPAK MedSystems
800-323-6305
www.corpakmedsystems.com
Multi-layer bag: nylon, ethylene vinyl
acetate, polypropylene
Non-DEHP PVC
Non-PVC bag
Non-DEHP tube
Kendall Healthcare
800-962-9888
www.kendallhq.com
Non-DEHP PVC
Non-DEHP bag & tube
Vygon
800-544-4907
www.vygonusa.com
Polyethylene
Extension set tubes
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 2 of 4)
Products
Manufacturer
Telephone
Webpage
Material
Comments
Enteral FeedingNasogastric (NG)
Tubes
CORPAK MedSystems
800-323-6305
www.corpakmedsystems.com
Silicone
Polyurethane
Gastotrostomy tube for neonates
PEG tube for neonates, nasoenteric
feeding tube
C. R. Bard, Inc.
800-545-0890
www.bardmedical.com
Silicone
Polyurethane
Nasogastric tube for neonates
Pediatric clear staright catheter
Kendall Healthcare
800-962-9888
www.kendallhq.com
Polyurethane
Nasogastric tube, PEG feeding tube
Kimberly-Clark
(Ballard Medical Devices)
800-524-3557
www.kchealthcare.com
Silicone
PEG feeding tube, gastrotomy feed
ing tube, jejunal feeding tube
Klein-Baker Medical
210-696-4061
www.neocare.com
Silicone
Feeding tube for neonates
Ross
800-231-3330
www.ross.com
Polyurethane
Nasoenteric feeding tube, nasojejunal feeding tube
Gastrostomy tube (some peds), PEG
tube
Silicone
Epidural Vessel
Catheters
Gloves, Examination
Utah Medical Products, Inc.
800-533-4984
www.utahmed.com
Silicone
Nasogastric and nasojejunal tubes
(neonates/peds)
Vygon
800-544-4907
www.vygonusa.com
Polyurethane
Silicone
Gastric feeding tubes for infants,
sump tube (Salem or Replogal)
Nasojejunal tubes
Zevex
800-970-2337
www.zevex.com
Polyurethane
Nasoenteric feeding tube
B. Braun
800-227-2862
www.bbraunusa.com
Polyamide (Nylon)
Epidural vessel catheter
Vygon
800-544-4907
www.vygonusa.com
Polyethylene, polyurethane or
polyamide (nylon)
Epidural vessel catheter
Allegiance Healthcare Corp.
800-964-5227
www.allegiance.net
Nitrile
Ansell-Perry
800-321-9752
www.ansellheallhcare.com
Nitrile
Best Manufacturing Co.
800-241-0323
www.bestglove.com
Nitrile
ECI Medical Technologies
902-543-6655
www.ecimedical.com
Styrene butadiene
Maxxim Medical
800-727-7951
www.maxximmedical.com
Polyurethane
S 3 3 I A 3 a
1 V 3 I a 3 W
d H 3 0
0 N V
3 A d
01
S 3 A I 1 V Nd 3 1 1 V
0
ALTERNATIVES
TO
PVC
AND
DEHP
MEDICAL
DEVICES
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 3 of 3)
Products
Manufacturer
Telephone
Webpage
Material
Gloves, Examination
(continued)
Safeskin Corporation
800-462-9993
www.safeskin.com
Nitrile
SmartCare Inc.
800-822-8956
www.smartcare.com
Nitrile
Tillotson Healthcare Corp.
800-445-6830
www.thcnet.com
Nitrile
B. Braun
800-227-2862
www.bbraunusa.com
Multi-layer bag: Polypropylene/polyethylene copolymer, polyester, elastomer
laminate
Polypropylene/polyethylene copolymer
Polyethylene
Intravenous (IV) Bags
and Tubing
Comments
IV bag (Excel)
IV bag (PAB)
IV set with PVC-free tube (no
longer manufacturing, but still
available from some vendors)
Budget Medical Products
800-569-1620
www.icumed.com
Non-DEHP PVC
IV tube
Children's Medical Ventures
800-377-3449
www.childmed.com
Non-DEHP PVC
IV administration sets
Curlin Medical
714-893-2200
www.curlinmedical.com
Non-DEHP PVC
Infusion tube
Office Supplies:
3-ring binders
•
Available from standard office
supply companies
800-847-0670
Patient ID Bracelets
Precision Dynamics Corp.
800-521-5123
www.pdcorp.com
Tyvek®
TabBand
800-940-3993
www.tabband.com
Tyvek®, polypropylene and polyethylene
Wristband & Medical Specialty
Products
800-348-6064
www.wristbandsupply.com
Tyvek®
Appropriate for short stays
Bivona Medical Technologies
800-847-8000
www.bivona.com
Silicone
Endotracheal tube, tracheostomy
tube
DHD Healthcare
800-553-5214
www.dhd.com
Silicone
Aerosol mask
Rusch
800-533-4984
wwvz.ruschinc.com
Red rubber or silicone
Reusable endotracheal tube
Utah Medical Products, Inc.
800-932-0760
www.utahmed.com
Co-polyester-polyethylene foam and
polypropylene
Disposable infant oxygen hood
Vital Signs
800-962-9888
www.vital-signs.com
Polyester
Oxygen or aerosol applicationsAero2Mask
Respiratory Therapy
Products
Polyethylene, cardboard
Appropriate for short stays
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 4 of 4)
Products
Manufacturer
Telephone
Webpage
Material
Sequential
Compression Device
Kendall Healthcare
800-846-3000
www.kendallhq.com
Polyolefins
Shower Curtains
Brookstone
800-222-6883
www.brookstone.com
Tyvek®
Nylon
Many manufacturers
Total Parenteral
Nutrition
Umbilical Vessel
Catheters
Urinary Catheters
Wound
Drains/Drainage
Systems
Comments
Abbott
800-766-1077
www.abbott.com
Non-DEHP PVC
Empty IV bag and tube
Baxter Healthcare, Fenwal Division
800-544-4907
www.baxter.com
Ethylene vinyl acetate
TPN bag
Vygon
800-962-9888
www.vygonusa.com
Polyurethane
Catheter for parenteral nutrition
and mid/long-term IV therapy
(See PICC lines above)
Kendall Healthcare
210-696-4061
www.kendallhq.com
Polyurethane
Umbilical vessel catheter
Klein-Baker Medical
800-533-4984
www.neocare.com
Silicone
Umbilical vessel catheter
(neonates)
Utah Medical Products, Inc.
800-544-4907
www.utahmed.com
Silicone or polyurethane
Umbilical vessel catheter
Vygon
800-545-0890
www.vygonusa.com
Polyurethane
Umbilical vessel catheter
C.R. Bard
800-658-0130
www.bardmedical.com
Polyurethane
Urethral catheter for pediatrics
Degania Silicone
401-658-0130
www.deganiasilicone.com
Silicone
Foley catheter
Klein-Baker Medical
800-533-4984
www.neocare.com
Silicone
Urinary drainage catheter
(neonates)
Utah Medical Products, Inc.
800-545-0890
www.utahmed.com
Silicone
Urinary catheters
C.R. Bard
401-658-0130
www.bardmedical.com
Silicone
Drains
Degania Silicone
800-533-4984
www.deganiasilicone.com
Silicone
Surgical and wound drains, tho
racic catheter, nephrostomy
catheter (may fit neonates)
www.utahmed.com
Silicone
Thoracic catheter
Utah Medical Products, Inc.
S 3 3 I A 3 a
1 V 3 I a 3 W
d H 3 0
0 N V
3 A d
01
S 3 A I 1 V N H 3 1 1 V
Health Care Without Harm does not endorse any or these products, has not test
ed them for safety or efficacy, and does not take responsibility for the accuracy of
the information or product performance. Listing here is based solely on informa
tion provided by the manufacturer. Non-PVC products may contain much smaller
amounts of DEHR Flexible PVC-free products still must be tested to ascertain
whether they are in fact DEHP-free. Products that contain latex and chlorine are
excluded from this table: latex products because of concerns over latex allergies
and chlorine containing products because of concerns over lifecycle hazards.
Exceptions are made for the few PVC products for which few or no non-PVC
products are available. In those cases non-DEHP products are identified. This
table is a work-in-progress.
Sources: Sustainable Hospitals Project, 2000, “Alternative Products,” see
http://sustainablehospitals.org (Lowell: Sustainable Hospitals Project, UMass
Lowell); and Tickner, Joel, et al, 1999, The Use of Di-2-EthylhexyI Phthalate in
PVC Medical Devices: Exposure, Toxicity, and Alternatives (Lowell: Lowell
Center tor Sustainable Production, UMass Lowell); and all information was veri
fied through telephone contacts with manufacturer representatives or review of
manufacturer website information.
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is paa of Coing Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit. or to find out how to get a complete kit.
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF certification nurk jnd term arc the x*k ptope m ec the Chlorine Free
PftvJiKU Asfaxuuun and are only ukJ
□uihunzed a nJ certified uicn
X
DEHP
Exposures
During the
Medical Care
of Infants
A Cause for Concern
Prepared by
Ted Schettler MD, MPH
Science and Environmental
Health Network
What is DEHP
(di-ethylhexyl phthalate)?
DEHP is one member of a family of
chemicals called phthalates and is used
as a plasticizer of polyvinyl chloride
(PVC) medical devices. Plasticizers provide PVC with flexibility, strength, and
bondability. Most PVC medical devices
contain 20-40% DEHP by weight, but
PVC tubing may contain up to 80%
DEHP1,2 DEHP-plasticized PVC prod
ucts are common in neonatal intensive
care units (NICUs). Manufacturers use
DEHP in bags that contain IV solutions,
enteral formula, and blood products, and
in tubing that delivers these fluids as
well as TPN and oxygen.
DEHP-containing PVC medical prod
ucts have been used for approximately
40 years. When rhe Food and Drug
Administration (FDA) was authorized
to regulate medical devices beginning
in the mid-1970’s, products made of
material formulations that had been
used previously were not tested to the
same degree as new products that
came to market after May, 1976. Any
change in the status of older products
or new products made of the same
material must be based on a FDA con
clusion that their use poses a signifi
cant risk of harm, rather than the
manufacturer being required to
demonstrate product safety.
How are patients exposed?
DEHP does not chemically bind to
PVC. DEHP may therefore leach from
plasticized PVC when a medical device
comes into contact with fluids, lipids,
and/or heat. DEHP is lipophilic and
leaches preferentially into lipid-containing solutions. The rate of DEHP
leaching also depends on storage condi
tions (e.g. temperature, contact time,
agitation).
In general, medical procedures that
require hours or days, like hemodialy
sis, blood transfusion, extracorporeal
membrane oxygenation (ECMO), total
parenteral nutrition (TPN), or enteral
feeding, result in higher DEFIP expo
sures than brief procedures. On a
weight basis, neonates in the neonatal
intensive care unit (NICU) are likely
to be among the most highly DEFIPexposed patients because of the regular
use of many different DEHP-contain
ing PVC products in that setting.
What are the health
effects of DEHP?
DEFIP is a reproductive and develop
mental toxicant in laboratory animal
testing. MEHR the monoester metabo
lite of DEHR is toxic to the Sertoli cells
of the testes, causing cellular abnormal
ities and impairing proliferation. In
rodents, developmental DEHP expo
sure causes more general adverse effects
on the structure and function of the
male reproductive tract.’ Effects on the
developing male reproductive tract
occur at far lower doses than are toxic
to adult animals.'5-6 The testicular tox
icity of DEHP has not been evaluated
in immature, prepubertal primates,
including humans. Based on develop
mental studies in animals, the Food and
Drug Administration (FDA) and the
National Toxicology Program’s Center
for Evaluation of Risks to Fluman
Reproduction conclude that some med
ical procedures result in DEFIP expo
sures that exceed the threshold
NOAEL (no observable adverse effects
level) in the developing male reproduc
tive tract (NOAEL by oral route of
exposure —3.7-14 mg/kg/day) and
exceed the FDA’s estimated tolerable
intake (TI), below which no adverse
effects are expected.7,8
In addition to effects on the develop
ing male reproductive tract, questions
have been raised about the effects of
DEFIP exposure on the liver and lungs.
One prospective study found cholesta
sis in infants supported by ECMO. '
The authors hypothesize that hemoly
sis during ECMO produces a large
bilirubin load, the excretion of which is
inhibited by inspissated bile and/or
un
<
LL.
U_
O
LU
or
<
CD
<
O
LU
s:
LU
DEHR Another study, however, did
not find cholestasis after ECMO,10 but
DEHP plasma concentrations in the
second study were substantially lower
than in the first (estimated aggregate
exposure levels 4.7'35 mg/kg vs. 42'
140 mg/kg). Recently, renewed concerns have surfaced about a contribu
tory role of DEHP in the genesis of
hepatotoxicity’ frequently observed in
infants receiving TPN.11 Although this
potential hazard has not been studied,
larger quantities of DEHP leach from
PVC tubing through which TPN solu
tion is passed than were previously
estimated. The authors of this study
estimate that infant exposures from
TPN may reach 10 mg/kg/day, which
is more than one order of magnitude
higher per kg than adult exposures
from hemodialysis, and are experi
enced daily.
X
o
err
ZD
o
un
LU
or
=>
CD
o
DEHP also leaches from PVC endotra
cheal tubes during use. One study doc
uments a direct relationship between
time of endotracheal tube use and
DEHP leaching.12 The authors
hypothesize a link between DEHP
exposure and the risk of bronchopul
monary dysplasia in premature new
borns. This potential hazard has never
been studied in infants. DEHP deposi
tion in the infant lung, however, has
been documented after ventilation
with PVC tubing.13
O-
X
LU
Q_
X
CD
The FDA also notes that DEHP leach
ing from PVC materials promotes
platelet aggregation and complement
activation, with the potential for
adverse clinical consequences, includ
ing microemboli.
Toxicokinetics of DEHP
Much of the DEHP administered via
die gastrointestinal tract is converted to
its monoester, mono-ethylhexyl phtha
late (MEHP), by intestinal lipases before
absorption into the systemic circulation.
In adult primates, including humans
and marmosets, a smaller proportion of
DEHP is hydrolyzed and absorbed as the
monoester [than in rats], apparently
because of less lipase activity in primate
intestine.15 The degree of biotransfor
mation of DEHP to MEHP is important
since MEHP is generally agreed to be
the testicular toxicant.
When DEHP is administered intra
venously, less DEHP is converted to
MEHP than if the exposure is via the
intestinal tract.16 In studies of patients,
including infants, undergoing hemo
dialysis or exchange transfusions, how
ever, significant levels of MEHP have
been measured in blood after these par
enteral exposures.1' In a study of 11
patients undergoing maintenance
hemodialysis for treatment of renal fail
ure, concentrations of the metabolite,
MEHR ranged from about 1/3 to 6
times the DEHP concentrations.18
These data demonstrate that a signifi
cant amount of DEHP is converted to
MEHP even after intravenous expo
sure to the parent compound.
Humans and primates largely excrete
the monoester via glucuronide conju
gation, whereas rodents further
hydrolyze MEHP into other interme
diates. 19 The glucuronidation path
ways of human children, however, do
not mature until they are 3 months
old.20 Thus, this important clearance
mechanism is not fully available to
neonates and young infants.
What are the levels of DEHP
exposure in the NICU?
Published reports of DEHP exposures
from various sources in the neonatal
intensive care unit are summarized in
the table. For critically ill neonates,
examining single sources of exposure
may substantially underestimate total
exposures. Babies who require ECMO,
for example, also require multiple
blood transfusions, parenteral feeding,
medications, and IV fluids. Breast milk
and enteral feeding formula may be
administered through DEHP-containing PVC tubing. Loff, et al. note that,
in infants receiving TPN, when infu
sions of other medications are also
administered, the load can easily reach
10 mg DEHP/kg/day.21 No studies
have quantified exposure to DEHP
from enteral feeding bags and tubing,
nasogastric tubes, breast milk pumps
and tubing, respiratory tubing, endo
tracheal tubes, oxygen masks, or all
sources combined.
Summary
Neonates and infants who receive med
ical care that includes the use of plasti
cized PVC products may easily be
exposed to DEHP at levels that are in
excess of the no observed adverse effect
level (NOAEL) in animal tests. For
some medical therapies, these exposures
also exceed the FDA-derived “tolerable
intake” (TI). (see table) The FDA has
concluded that total parenteral nutri
tion, enteral feeding, exchange transfu
sions, and ECMO can individually
i
result in DEHP exposures that exceed
the TI by 3-50 fold. Of course, multiple
simultaneous medical procedures using
DEHP-containing PVC products will
more readily result in exposures in
excess of the TI. DEHP toxicity in the
developing male reproductive system is
the greatest known risk, with additional
concerns about thrombus formation,
microemboli, and impacts on the liver
and lungs. An expert panel convened
by the National Toxicology' Program’s
(NTP) Center for Evaluation of Risks to
Human Reproduction concluded that:
“[for DEHP] the available reproductive
and developmental toxicity data and
limited but suggestive human exposure
data indicate that exposures of inten
sively-treated infants and children can
approach toxic doses in rodents, which
causes the panel serious concern that
exposure may adversely affect male
reproductive tract development.”
The panel also expressed “concern
that ambient oral DEHP exposures
[primarily from general dietary con
tamination] to pregnant or lactating
women may adversely affect the devel
opment of their offspring.” DEHP
exposures from medical therapy would,
of course, add to ambient, dietary
exposures. The FDA characterizes
their safety assessment of DEHP as
entirely consistent with the NTP
panel’s concerns.
Potential exposures to DEHP from medical procedures and nutrition
m
in a neonatal intensive care unit
Source of
DEHP Exposure
Exposure
(mg DEHP/kg
body weight)
Unit
Artificial ventilation in preterm
infants (PVC respiratory tubing;
not polyethylene)
NR
Hour
(inhalation)
Neonatal blood replacement
transfusion; short-term, acute
0.3 (0.14-0.72)
treatment
period
NR
2
2
Neonatal blood replacement
transfusion; double volume; short
term, acute
1.8 (0.84-3.3)
treatment
period
NR
3
0.3
Platelet concentrates in newborns
1.9
treatment
NR
4
0.3
Extracorporeal oxygenation in
infants
14-140
treatment
NR
5
0.04-0.004
Extracorporeal oxygenation in
infants
4.7-34.9
Treatment
NR
6
0.12-0.02
1-4 hours
0.3-4.7
mg/mL/hr(change in
level in whole blood
during procedure)
7
NR
8
Total Exposure or
Concentration in
Product
Source
Tl/dose*
tn
0.001-4.2 mg(est.
total exposure)
1
m
Congenital heart repair
(neonates)
•
20
0.03
From tubing
Total parenteral nutritional
formula (TPN), with lipid
2.5
NR
3.1 ug/mL (concentra
tion in TPN formula);
more from tubing
9
0.2
TPN/IV Tubing
5
day
10 mg/2-kg baby/day
10
0.12
Multiple IV Sources: packed red
blood cells, platelet rich plasma,
fresh frozen plasma, and medications
5
day
10 mg/2-kg baby/day
11
0.12
Breast milk
0.0015-0.0165
Day
0.01-0.11 mg/kg (con
centration in breast
milk)
12
27-2.4
Infant formula
0.015
Day
0.004-0.06 mg/kg wet
weight
13
2.6
Infant formula
0.0087-0.035
NR
0.33-0.98 mg/kg dry
weight
14
4.5-1.1
IV crystalloid solution
m
•
co
Reported *TI/dose- based on FDA's TI of 0.6 mg/kg/day for parenteral exposures and 0.04 mg/kg/day for intestinal
exposures; Tl/dose ratios < 1 imply that the TI has been exceeded for the given source of exposure
3
Notes
1.
NTP-CERHR expert panel report on di(2-ethylhcxyDphthalate. National Toxicology
Program. US Dept of Health and Human
Services; Oct. 2000.
2.
DiGangi J. Phthalates in vinyl medical prod
ucts Washington DC: Greenpeace USA, 1999.
3
Gray E, Wolf C, Lambright C, et al.
Administration of potentially anriandrogenic
pesticides (procymidone, linuron, iprodione,
chlozolinate, p,p’-DDE, and ketoconazole) and
toxic substances (dibutyl- and diethylhexyl
phthalate, PCB 169, and ethane dimethane
suphonate) during sexual differentiation pro
duces diverse profiles of reproductive malfor
mations in the rat. Toxicol Ind Health 14: 94118, 1999.
4.
5.
6.
7.
Lamb J, et al. 1987. Reproductive effects of
four phthalic acid esters in the mouse. Toxicol
Appl Pharmacol 88. 255-269.
Arcadi R, Costa C, Imperatore C, et al. Oral
toxicity of DEHP during pregnancy and suck
ling in the Long-Evans rat Food Chem Toxicol
36:963-970, 1998.
Poon R, Lecavalier R Mueller R, et al.
Subchronic oral toxicity of di-n-ocryl phthalate
and DEHP in the rat. Food Chem Toxicol
35:225-239, 1997.
N'TP-CERHR expert panel report on di(2-ethylhexyl)phthalate. National Toxicology
Program US Dept of Health and Human
Services; Oct, 2000.
8.
US FDA Safety assessment of di(2-ethylhexyl)phthalate (DEHP) released from PVC
medical devices. Sept, 2001.
9.
Schneider B, Schena J, Truog R, et al. A
prospective analysis of cholestasis in infants
supported with extracorporeal membrane oxy
genation J Pediatr Gastroenterol Nurr 1 3: 28589, 1991.
10. Karie V, Short B, Martin G, et al. Extracorporeal
membrane oxygenation exposes infants to the
plasticizer, di(2-ethylhexyl)phthalate Cut Care
Med 25:696-703, 1997.
15. Pollack G. Li R, Ermer J, et al Effects of route
of administration and repetitive dosing on the
disposition kinetics of cii(2-ethylhexyl) phtha late and its mono-de-estenfied metabolite in
rats. Toxicol Appl Pharmacol 79:246-256,
1985.
16. Rubin R, Schiffer C. Fate in humans of rhe
plasticizer, di-2-ethylhexyl phthalate, arising
from transfusion of platelets stored in vinyl
plastic bags. Transfusion 16:330-335, 1976.
17. Sjoberg R Bondesson U, Sedin E, et al.
Exposure of newborn infants to plasticizers.
Plasma levels of di-(2-ethylhexyl) phthalate
and mono-(2-ethylhexyl) phthalate during
exchange transfusion. Transfusion 25 (5).424428, 1985.
18. Pollack G, Buchanan J, Slaughter R, et al.
Circulating concentrations of di(2ethylhexyl)phthalate and its de-esterified
phthalic acid products following plasticizer
exposure in patients receiving hemodialysis.
Toxicol Appl Pharmacol 79:257-267, 1985.
19. Albro R Corbetr J, Schroeder J, et al.
Pharmacokinetics, interactions with macromol
ecules and species differences in metabolism of
DEHP Environ Health Perspect 45:19-25,
1982.
20. Creistel T Onset of xenobioric metabolism in
children toxicological implications. Food
Addit Contain 15:45-51, 1998.
21. Loff S, Kabs F, Witt K, et al. Polyvinylchloride
infusion lines expose infants to large amounts
of toxic plasticizers J Pediatr Surgery' 35(12);
1775-1781,2000.
13. Roth B, Herkenrath P Lehmann H, er al. Di(2-ethylhexyl)-phthalate as a plasticizer in
PVC respiratory tubing systems: indications of
hazardous effects on pulmonary function in
mechanically ventilated, preterm infants. Eur J
Pediatr 147: 41-46, 1988.
14. US FDA. Safety assessment of di(2-ethylhexyl)phthalate (DEHP) released from PVC
medical devices. Sept, 2001
Loff, S, Kabs F, Witt K, Sartoris J, et al.
Polyvinylchloride infusion lines expose infants
to large amounts of toxic plasticizers, J Ped
Surg, 35: 1775-1781, 2000.
9.
Mazur HI, Stcnnett DJ, and Egging PK.
Extraction of diethylhexylphthalate from total
nutrient solution-containing polyvinyl chloride
babs. J Parenter Enter Nutt, 13.59-62, 1989.;
Loff, S, Kabs F, Witt K, Sartoris J, et al.
Polyvinylchloride infusion lines expose infants
to large amounts of toxic plasticizers, J Ped
Surg, 35: 1775-1781,2000.
10. Loff, S, Kabs F, Witt K, Sartoris J, et al.
Polyvinylchloride infusion lines expose infants
to large amounts of toxic plasticizers, J Ped
Surg, 35: 1775-1781, 2000.
11. Loff, S, Kabs F, Witt K, Sartoris J, et al.
Polyvinylchloride infusion lines expose infants
to large amounts of toxic plasticizers, J Ped
Surg, 35: 1775-1781, 2000.
12. Pfordt J and Bruns-Weller E. 1999 Die
Phthalsiiurecster als eine Gruppe von Umweltchemikalien nnt endokrinen Potential.
Niedarsachsisches Ministerium fhr Ernahrung,
Landwirschaft und Forsten.
13 Petersen J and Breindahl T. Plasticizers in total
diet samples, baby food, and infant formulae,
Food Additives and Contaminants, 17; 133141, 2000.
14- MAFF. Food surveillance information sheet Phthalates in infant formulae. Joint Food
Safety' and Standards Group: MAFF - UK,
1996.
Table Sources
1
Roth B, Herkenrath R Lehman H, et al. Di-(2ethylhexyl)-phthalate as plasticizer in PVC res
piratory' tubing system; indications of hazardous
effects on pulmonary function in mechanically
ventilated, preterm infants. J Pediatr 147:4146, 1988.
2
Sjoberg R Bondesson U, Sedin E, et al.
Exposure of newborn infants to plasticizers:
Plasma levels of di-(2-ethylhexyl) phthalate
and mono-(2-ethylhexyl) phthalate during
exchange transfusion. Transfusion 25(5):424428, 1985.
3.
Sjoberg, 1985.
4-
Huber WW, Grasl-Kraupp B, and SchulteHermann R. Hepatocarcmogenic potential of
DEHP in rodents and its implications on
human risk, Critical Reviews in Toxicology, 26:
365-481, 1996.
11. Loff S, Kabs F, Witt K, et al. Polyvinylchloride
infusion lines expose infants to large amounts
of toxic plasticizers. J Pediatr Surgery 35(12):
1775-1781,2000.
12. Latim G, Avery G. Materials degradation in
endorrachael tubes: a potential contributor to
bronchopulmonary dysplasia. Acta Pediatr
88(10).1174-5, 1999.
8.
5.
Schneider B, Schena J, Troug R, et al. Exposure
to di(2-ethylhexyl)phthalate in infants receiv
ing extracorporeal membrane oxygenation.
New Engl J Med 320:1563, 1989.
6.
Karie VA, Short Bl, Martin GR et al.
Extracorporeal membrane oxygenation exposes
infants to the plasticizer, DEHR Critical Care
Medicine, 25: 696-703, 1997.
7.
Barry YA, Labow RS, Keon, WJ, et al.
Perioperative exposure to plasticizers in
patients undergoing cardiopulmonary bypass.
J Thorac Cardiovas Surg, 97: 900-905, 1989.
HeaLthfCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.nohann.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
rhe PCF certincahan mark and term are the sole property of the Chlorine Free
Products Association and are only uu-d by authorized and certified
the Expert
Panel Report
of the
National
Toxicology
Program on
DEHP and its
•Risks to
Human
Reproduction1
In October 2000, the National
Toxicology Program of the U.S.
Department of Health and Human
Services’ Center for the Evaluation of
Risks to Human Reproduction (NTPCERHR) released an Expert Panel
report on Di(2-ethylhexyl)phthalate
(DEHP) and its risks to human devel
opment and reproduction. Below is a
summary of the Expert Panel's findings.
The Panel did not consider risks other
than those to reproduction and devel
opment. The NTP has not yet
released its official findings.
Overall Findings
The National Toxicology' Program’s
Expert Panel report focuses concern on
three distinct populations at risk of
DEHP exposure: critically ill infants,
healthy infants and toddlers, and the
offspring of pregnant or lactating
women.
Critically ill infants: “The available
reproductive and developmental toxici
ty data and the limited but suggestive
human exposure data indicate that
exposures of intensively-treated
infants/children can approach toxic
doses in rodents, which causes the
Panel serious concern that exposure
may adversely affect male reproductive
tract development.”2
Healthy infants and toddlers: “If
healthy human infant/toddler exposure
is several-fold higher than adults, the
Panel has concern that exposure may
adversely affect male reproductive tract
development.”'
Pregnancy and lactation: “[T]he
panel has concern that ambient oral
DEHP exposures to pregnant or lactat
ing women may adversely affect the
development of their offspring.”1
DEHP: Preferred
Plasticizer for
Medical Devices
Di(2-ethylhexyl)phthalate, commonly
referred to as “DEHR” is used as a plas
ticizer of polyvinyl chloride (PVC) in
the manufacture of a wide variety of
consumer products. Plasticizers provide
PVCs with characteristics such as flexi
bility, strength, and bondability.
Plasticizers allow PVC to be softened
and shaped into many designs without
cracking or leaking, “an important per
formance characteristic for medical
devices.”5
DEHP is currently the only phthalate
plasticizer used in PVC medical
devices. DEHP is used as a plasticizer
for medical devices because it can pro
vide the “desired mechanical proper
ties” to the PVC.6 By weight, PVCbased medical devices contain, on
average, 20%-40% DEHP'
Medical Procedures
Where PVCs are Most
Commonly Used
A variety of medical procedures use
PVC-containing devices including:
administration of intravenous (IV) flu
ids; cardiopulmonary' bypass; ECMO
(extracorporeal membrane oxygena
tion); enteral and total parenteral
nutrition feedings; hemodialysis; respi
ratory therapy; and transfusion of
whole blood, platelets, or plasma?
PVC tubing is used in medical applica
tions such as extracorporeal membrane
oxygenation (ECMO), feeding tubes,
hemodialysis, IV fluid tubing, and
mechanical ventilation.9
Absorption and
Metabolism of DEHP
DEHP administered via the gastroin
testinal tract is substantially converted
to the monoester, MEHR by intestinal
lipases before absorption into the sys-
temic circulation. “In primates, including humans and marmosets, a smaller
proportion of DEHP is hydrolyzed and
absorbed as the monoester [than in
rats], apparently because of less lipase
activity in primate intestines.”10 When
DEHP is administered intravenously,
“the ratio of DEHP to its monoester in
blood is much higher than if the DEHP
is received orally.”11 The degree of biotransformation of DEHP to MEHP is
important since MEHP is generally
agreed to be the testicular toxicant
that has led NTP-CERHR to raise a
“serious concern” for neonates. (See
“Development and Reproductive
Effects of DEHP” section below.)
According to NTP-CERHR’s Expert
Panel, “(i]n a study of 11 patients
undergoing maintenance hemodialysis
for treatment of renal failure....... con
centrations of the metabolite, MEHR
ranged from about 1/3 to 6 times the
DEHP concentrations.”12 These data
demonstrate that a significant amount
of DEHP is converted to MEHP even
after intravenous exposure to the par
ent compound. Humans and primates
largely excrete the monoester as a glu
curonide, whereas rodents further
metabolize this intermediate.” The
glucuronidation pathways of human
children, however, do not mature until
they are 3 months old. Thus, the
important clearance mechanism is not
fully available to neonates and young
infants.”14
Developmental
and Reproductive
Effects of DEHP
The Expert Panel only looked at the
risks of DEHP exposure to human
reproduction and development.15
Numerous animal studies indicate that
the liver and testes are target organs of
DEHR “ [I] t is clear from the existing
data that testicular pathology and
reduced sperm numbers are consistent
effects [of DEHP].”16 Testes, and
specifically the Sertoli cell, have been
identified as a target.17 “The data are
sufficient to conclude that DEHP is a
reproductive toxicant in male rats,
mice, ferrets, and guinea pigs when
administered orally.”1'’
IT] here are sufficient data in rodents
to conclude confidently that oral expo
sure to DEHP can cause reproductive
and developmental toxicity in rats and
mice. Further, an effect observed in
rats involves adverse effects on the
development, structure, and function
of the male reproductive tract. Thus,
for DEHR the effects on reproduction
and development are intertwined.1’
Because reproductive effects occur at
lower doses than are toxic to adult ani
mals, “current concern focuses on pre
natal exposure leading to postnatal
toxicity.”20 As the NTP-CERHR
Expert Panel report indicates,
[R]ecent mechanistic studies have doc
umented that phthalates are more
potent reproductive toxicants at lower
doses when exposure occurs during
gestation. The most sensitive end
points are those that monitor the
development and formation of the
reproductive system, testes descent,
prepuce separation (also known as balanopreputial separation) in males, and
vaginal opening and onset of estrous
cycling in females.’1
“Based on these studies, it would
appear from the current data set that
the LOAEL (lowest observed adverse
effect level) is —38 mg/kg bw/day and
the NOAEL (no observed adverse
effect level) is —3.7-14 mg/kg bw/day
for reproductive effects in rodents by
the oral route.”22
The NTP-CERHR Expert Panel report
describes apparent species differences
in the reproductive toxicity of DEHP
and its metabolites. Two studies of
juvenile or pubertal non-human pri
mates (marmosets and cynomolgus
monkeys) showed no effect on testicu
lar weight after 2-13 week oral expo
sures to DEHP at 500-2500 mg/kg/day.
The Panel concluded that for the
cynomolgus monkeys,
It the cynomolgus monkey were as sen
sitive as a juvenile rat to the effect of
DEHR testicular histopathology would
have been observed. If the monkeys
were only as sensitive as an adult rat,
this dose would have been ineffective
in producing testicular toxicity. Thus,
while this study is useful in confirming
that monkeys are not as sensitive as
the most vulnerable of other model
species, it is not useful in confidently
placing the monkey along the spectrum
of susceptibility to DEHP-induced tes
ticular damage. As such, it is of limit
ed use to the Panel in determining the
likely risk of DEHP to human repro
duction.2’
Furthermore, there is “moderate-tolow” confidence that the authors
found the real NOAEL or LOAEL for
marmosets, because the most suscepti
ble age of animal was not exposed and
the most sensitive endpoints were not
examined.24
I
Exposure to DEHP
Can Be Increased Under
Certain Conditions
The NTP-CERHR Expert Panel
reports that DEHP does not bind to
PVC, but instead leaches out when
the medical device comes in contact
with fluids or is heated.
Since the DEHP plasticizer is not
chemically bound to PVC, it can leach
out when the medical device contacts
fluids such as blood, plasma, and drug
solutions, or it can be released and
migrate when the device is heated.
The rate at which DEHP migrates from
the medical device into the stored
material depends on the storage condi
tions (temperature of the fluid contact
ing the device, the amount of fluid, the
contact time, the extent of shaking or
flow rate of the fluid) and the
lipophilicity of the fluid.25
DEHP leaches into many IV and
enteral formulas/solutions, including
whole blood, plasma, total parenteral
and enteral nutrition solution, and
solutions containing Polysorbate 80
and other formulation aids used to solubize some IV medications.20 DEHP
leaching also may occur during sterili
zation and irradiation.27 The condi
tions under which medical devices are
stored or treated can increase the
migration of DEHR Hence, “[l|ong
storage or use time, increased temper
ature, and agitation all increase leach
ing out of DEHP from medical
devices. Leaching is also enhanced by
increased lipid content or by the
lipophilic nature of liquids that con
tact DEHP in medical devices.”2'
In general, medical procedures that
require hours or days, like hemodialysis
or ECMO, result in higher DEHP
exposures than brief medical proce
dures like infusion of packed red blood
cells or administration of IV medica
tions. Chronic or recurrent treatments
like hemodialysis in chronic renal fail
ure patients or multiple, long-term
transfusions in cancer victims can
result in cumulatively high exposures.
Intensive procedures like exchange
transfusions in neonates can result in
acutely high exposures.2"
Neonate exposures to DEHP from
exchange transfusions range from
1,700-4,200 ug/kg bw per treatment.,-?
Two studies estimated neonate expo
sures from ECMO at 42,000 to
140,000 ug/kg bw (or 4,200 to 14,000
ug/kg bw/day); and non-detect (using
heparin-coated tubes) to 34,900 ug/kg
bw per treatment (or non-detect to
3.49 ug/kg bw/day).'1 Evidence indi
cates that coated tubes may not leach
as readily as non-coated.
IV Administration of
Solutions and Drugs
IV administration of solutions and
^rlrugs may result in exposure to DEHP
-^^[S]everal studies have found that if
normal saline and glucose solutions in
PVC bags are agitated, DEHP may
form an emulsion, increasing the
amount of DEHP extracted into the
solutions.A variety of drugs are
administered intravenously by adding
them to PVC IV bags. Although phar
maceutical solvents such as ethanol
and polyethylene glycol do not affect
the extraction of DEHP from PVC
storage bags, formulation aids such as
Polysorbate 80 and castor oil dramati
cally increase the rate of DEHP
extraction.3’ In addition, some drug
formulations significantly increase the
extraction of DEHP from the PVC
container into the solution.34
Total Parental Nutrition
The NTP-CERHR Expert Panel report
recognizes that one of the greatest
risks of DEHP exposure exists in total
parental nutrition (TPN) formulations.
[TPN| formulations contain amino
acids, dextrose, electrolytes, and lipids.
The presence of lipids have been
shown to increase extraction of DEHP
from PVC bags. In TPN formulations
without added lipids, there was no
measurable amount of DEHP In TPN
formulations with added lipids, the
concentration of DEHP in the TPN
solutions increased with time and stor
age temperature.35
Single v. Multiple
Exposures to DEHP
According to the NTP-CERHR Expert
Panel,
For many patients, particularly critical
ly ill neonates, examining single
sources of exposure (e.g., ECMO or
ventilation) may substantially under
estimate DEHP exposure. Babies who
require ECMO, for example, also
require multiple replacement blood
transfusions, parenteral feeding, med
ications, and IV fluids. Many of these
other inputs could substantially
increase DEHP exposure. DEHP also
passes through breast milk which,
when available, is used in some criti
cally ill, hospitalized babies, as part of
enteral nutrition.”’
Hence, multiple sources of DEHP
exposure must be considered when
evaluating the aggregate risk to an
individual patient in a medical care
setting.
mental toxicity data and the limited
but suggestive human exposure data
indicate that exposures of intensivelytreated infants/children can approach
toxic doses in rodents.'8 Thus, the
Panel concluded that pediatric expo
sure represents a special case.” Once
again, NTP only reviewed the repro
ductive and developmental toxicity
data of DEHP
Exchange transfusions, ECMO, and
cardiopulmonary by-pass for correction
of congenital anomalies all represent
high exposure scenarios.... ECMO,
used for refractory respiratory7 failure in
both premature infants and term
infants, gives one of the highest single
course exposures to DEHR40
The scientific evidence available to
the NTP-CERHR Expert Panel lead it
to raise serious concerns about neona
tal DEHP exposure and its potential
link to reproductive problems. The
report finds that:
D “(I]nfants undergoing routine
replacement blood transfusions
may be exposed to doses of DEHP
1-2 orders of magnitude above gen
eral population exposures and have
concomitant MEHP exposure.”41
(MEHP is the monoester metabo
lite of DEHR);
D
“DEHP levels in necropsy tissues
(heart and gastrointestinal) from
premature neonates who received
varying quantities of blood prod
ucts were found to be significantly
higher in comparison to those of
infants who had not received blood
transfusions;”42
B
“Infants undergoing intensive ther
apies may be exposed to levels up
to 3 orders of magnitude above
general exposures. Chronic expo
sures in adults undergoing
hemodialysis can be 1-2 orders of
magnitude above average exposure
to DEHP;”43
■
“Documented parenteral medical
exposure to DEHP of critically ill
infants can exceed general popula
tion exposures by several orders of
magnitude;” 44
Pediatric DEHP Exposure,
Particularly in Neonates,
Raises "Serious Concern"
While the NTP Expert Panel recog
nized that the benefits of medical pro
cedures can outweigh any risks, the
Expert Panel had “serious concern”
that DEHP exposure may adversely
affect male reproductive tract develop
ment of critically ill infants.37 The
available reproductive and develop
“{MJedical exposures from simultane
ous interventions in the same patient
(e.g., ventilation, nasogastric tubes,
transfusion, and parenteral feeding)
have not been quantified. Such
exposures may result in dose levels
considerably above those document
ed for single medical procedures;”45
and
18. Page 94- “The oral repeat-dose studies in rats
and mice consistently show that the primary
targets for effect are liver, kidney, and testes.
Effects were also observed in some studies on
the pituitary, thyroid, thymus, ovaries, and
blood. While the liver shows a biological
response at the lowest doses of DEHP that
cause effects, the testes’ response at somewhat
higher doses is a greater health hazard con
cern.” Page 80.
39. Second page 12. “Several important exposures
in the fetus and neonates have not been
explored, including placental transfer of mater
nally-derived DEHP/MEHP from medical
and/or dietary' sources, and contributions from
parenteral and enteral feeding, ventilators, IV
fluids, or combinations of simultaneous expo
sures. It is likely that such investigation would
yield higher exposures to small babies during a
developmentally vulnerable time.” Page 79.
19. Page 99.
40. Second page 12.
‘‘The 3-30 ug/kg/d range of exposure
for the general population may be
increased by 2-3 orders of magnitude
for infants undergoing intensive ther
apeutic interventions.”'0
20. Page 72.
41. Page 98.
21. Page 60.
42. Page 34.
22. Page 101.
43. Page 102.
23. Page 67
44. Page 101.
24. Page 66.
45. Page 102.
25 Page 10.
46. Page 101.
26. Page 10.
Notes
27. Page 10.
1.
Source document: NTP-CERHR Expert Panel
Report on Di(2-ethylhexyl)phthalate, National
Toxicology Program, U.S. Department of
Health and Human Services, Center for the
Evaluation of Risks to Human Reproduction,
October 2000. Note, the downloadable NTPCERHR report that is available from CERHR
by mail or the internet site has double pages 912. Hence, all cites to the four repeated pages
will be identified as “second page__ .”
28. Second page 9.
2.
Page 101.
3.
Page 101.
4.
Page 102.
5.
Page 9.
6.
Page 9. “These mechanical properties include
flexibility, strength, suitability for use at a wide
range of temperatures, suitability for various
sterilization processes, resistance to kinking,
optical clarity, weldability, barrier capability,
centrifugability, and bondability.”
7.
Page 10.
8.
Pages 9-10.
9.
Page 12.
34. Page 12. Other drug formulations that signifi
cantly increase the leaching of DEHP include,
cefoperazone (Cefobid Bulk), chlordiazepoxide
HCL (Librium), ciproflocaxin (Cipro IV),
cimetidine (Tagamet), cyclosporine
(Sandimmune), etoposide (VePesid), flucona
zole (Diflucan), metronidazole HC1 (Flagyl
IV), micronazole (Monistar IV), paclitaxel
(Taxol), tracrolimus (Prograf), taxotere
(Docetaxel), teniposide (Vumon), total par
enteral nutrition formulas, and vitamin A.
“The highest DEHP concentrations are
reached when the drugs are pre-mixed in IV
bags and the premixed solution is agitated for
24 hours. However, it should be noted that
clinicians and nurses are familiar with the
increased leaching.of DEHP from PVC con
tainers into lipophilic drug formulations.
Pearson and Trissei have recommended that
these drug formulations be prepared in nonPVC containers and administered through
non-PVC tubing. The labeling of such formu
lations includes a warning to that effect.”
10. Page 85.
11. Page 34.
12. Page 34.
13. Page 85.
14- Page 85.
15. In addition to effects on the developing male
reproductive tract, questions have been raised
about the effects of DEHP exposure on the
liver and lungs.
16. Page 94.
17. Page 98.
29. Page 13.
30. Second page 12.
31. Second page 12.
32. Page 11. “As expected, very little PVC leaches
into normal saline solutions form PVC storage
bags even alter long periods of storage.”
33. Page 12.
35. Page 12.
36. Page 13.
37. Page 101.
38. Page 101.
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
Die Pci- certification mark and term arc the sole property of the Chlorine Free
Products /Xuociation and arc (inly u*cd by authorued and certified usen.
Resources
on PVC and
DEHP in
Health Care
US Government Publications
U.S. Food and Drug Administration
(FDA), Center for Devices and
Radiological Health. 2001. Safety
.Assessment of Di (2-ethylhexyl) Phthalate
(DEHP) Released from PVC Medical
Devices. Rockville, MD: U.S. FDA.
Other Materials on PVC or DEHP
in Healthcare
Health Care Without Harm. 2001.
Dioxin, PVC and Health Care
Washington, DC: Health Care Without
Harm. Webpage: www.noharm.org.
Health Care Without Harm. 2001.
National Toxicology Program, Center for
the Evaluation of Risks to Human
Reproduction (CERHR). 2000. NTP
CERHR Expert Panel Report on
Di (2-ethylhexyl) Phthalate. Webpage:
http://cerhr.nielas.mh.gov/news/index html
United State Environmental Protection
Agency (US EPA). 2000. Draft Exposure
and Human Health Reassessment of
2,3,7,8'Tecrachlorodibenzo-p'Dioxin
(TCDD) and Related Compounds.
Washington, DC: US EPA. Webpage:
http.//www.epa.gov/ncea/pdfs/dioxm/part
land2.htm.
European Government
Publications
Swedish National Chemicals
Inspectorate. 2000. Risk Assessment:
bis(2'ethylhexyl) phthalate (Final Draft).
Soina, Sweden.
Danish Ministry’ of Environment and
Energy. 1999. Action Plan for Reducing
and Phasing Out Phthalates in Soft Plastics.
Copenhagen, Denmark.
European Commission. 2000. Green
Paper on Environmental Issues of PVC.
Webpage: www.europa.eu.int/
comm/environment/pvc/index.htm
European Commission. 2000.
Five PVC studies:
1. The Influence of PVC on the Quantity
and Hazardousness of Flue Gas
Residues from Incineration
2. Economic Evaluation of PVC Waste
Management
3. The Behaviour of PVC in Landfill
4. Chemical Recycling of Plastics Waste
(PVC and Other Resins)
5. Mechanical Recycling of PVC Wastes
Webpage: www.europa.eu.int/comm/
environment/waste/facts en.htm
Reducing PVC Use in Hospitals.
Washington, DC: Health Care Without
Harm. Webpage: www.noharm.org.
Health Care Without Harm. 2001. A
Summary of the FDA Safety Assessment of
DEHP Released from PVC Medical
Devices. Washington, DC: Health Care
Without Harm. Webpage:
www.noharm.org.
Rossi M. 2000. Neonatal Exposure to
DE HP and Opportunities for Prevention.
Falls Church, VA: Health Care Without
Harm. Webpage: www.noharm.org.
Rossi M, Schertler T. 2000. “PVC
White Paper.’’ In Proceedings from Setting
Healthcare's Environmental Agenda (San
Francisco, CA). Falls Church, VA:
Health Care Without Harm.
Schertler T. 1999. Do We Have a Right
to Higher Standards? C. Everett Koop, MD
and an ACSH Panel Review the Toxicity
and Metabolism of DEHP Falls Church,
VA: Health Care Without Harm.
Webpage: www.noharm.org.
Schertler T. 2001. DEHP Exposures
During the Medical Care of Infants: A
Cause for Concern. Washington, DC:
Health Care Without Harm. Webpage:
www.nohami.org.
Silas J. 2001 A Summary of the Expert
Panel Report of the National Toxicology
Program on DEHP and its Risks to Human
Reproduction. Washington, DC: Health
Care Without Harm. Webpage:
www.noharm.org.
Tickner J, Schertler T, Guidotri T, McCally
M, Rossi M. 2001. “Health Risks Posed by
Use of Di-2-Ethylhexyl Phthalate (DEHP)
in PVC Medical Devices: A Critical
Review.” American Journal of Industrial
Medicine. 39:100-111.
Tickner J, Hunt R Rossi M, Haiama N,
Lappe M. 1999. The Use of Di-2-
Ethylhexyl Phthalate in PVC Medical
Devices: Exposure, Toxicity, and
Alternatives. Lowell: Lowell Center for
Sustainable Production, University of
Massachusetts Lowell. Webpage:
www.nohami.org.
University of Massachusetts Lowell,
Sustainable Hospitals Project. 2000.
“Alternative Products." Webpage:
www sustainablehospitals.org.
Video: “First Do No Harm: PVC and
Medicine’s Responsibility." Western
Lake Superior Sanitary District, MN.
(2000). (For copies contact Health Care
Without Harm at hcwh@chej.org or
202-234-0091).
Health rCare')
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.nohami.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit or to find out how to get a complete kit.
visit Health Care Without Harm on the Web at www.noharm.org .
The PCF cc ruck acion mark and term are (he wk property <jf (he Chlorine Free
Produce* Aitccuttun and are only u>cd K luthoriard and certified ukc*.
Waste
Minimization,
Segregation
and Recycling
in Hospitals
There isn’t a healthcare organization
anywhere that does not strive to
improve patient satisfaction; delivery' of
care; performance as a corporate citi
zen; and the bottom line. Even the
best endowed among nationally promi
nent health care providers have sought
to soften the blow of decreasing
Federal dollars.
tal services, someone from the
product selection/safety team, risk
managers, safety managers, director
of nursing, and interested employ
ees and staff. This group can then
strategize about courses of action
for the facility with input from all
responsible sectors.
In 1996, Beth Israel Medical Center in
New York City implemented an aggres
sive waste minimization plan that
sought to minimize both the volume
and the toxicity of the waste their
facility generated. As a result, they
continue to save over $600,000 a year.
Here are some beginning steps any
health care organization can take to
minimize waste.
2. Conduct a waste audit. It doesn’t
have to be done by a consultant, it
can be handled by a nurse or an
employee from environmental serv
ices. Take a good look at everything
that is coming into your hospital
(through the Purchasing /Materials
Management Department) to
everything that exits the hospital—
in the form of recyclables, red bag
waste, solid waste, food waste, labo
ratory' chemicals, chemotherapeutic
and pathological waste. You may be
surprised to find that about 85% of
the waste that exits the hospital is
non-infectious waste similar to that
you’d find in a large hotel or office
building. The chart bellows illus
trates the composition of hospital
waste.
1. Establish a “Green Team.”
Convene a task force of administra
tors, housekeepers, nurses and oth
ers who are responsible for waste
handling. You can add your direc
tors of purchasing and environmen
3. Waste segregation is an important
step in reducing the volume of
waste, because it offers the ability'
to make more accurate assessments
about the composition of the hospi
tal’s waste, and positions the facili
ty for different management
Everyone is asked to look for a way to
cut potential costs within the hospital
environment. Here are several strategies
that can not only save facilities thou
sands of dollars, they also significantly
lessen hospitals’ impact on the environ
ment and community around them.
Hospital Solid Waste Composition
Metals
Other
12%
Paper
53%
o
o
r-
17%
strategies.1 Use the results from the
waste audit to identify wasteful
practices and design a waste man
agement strategy that incorporates
waste reduction, reuse, and recy
cling measures.
4. Education is a top priority. Teach
nursing and housekeeping staff the
proper way to segregate waste.
Train staff about the environmental
consequences of medical waste
incineration. Post signs where waste
is sorted.
5. Recycling. Don’t throw out what
you can recycle. Make recycling a
priority. There are more than 25
materials in a hospital that can be
safely and easily recycled.
Cardboard, glass, office paper,
drink cans, newspapers, magazines,
and PETE #1 and HPDE #2 plas
tic have nationwide recycling mar
kets. Set aside space for bins and
work with your waste hauler to
expand the scope of your recycling
contracts.
6. Purchasing practices are key in
pursuing aggressive waste minimiza
tion. Work with your purchasing
team to select reusable rather than
disposable products. Have your
product selection team examine the
environmental impacts/safety of
materials coming into the hospital.
Work with your risk manager to
choose products that don’t have a
negative impact on worker or
patient health and safety.
Implement a purchasing program
that favors products made of recy
cled paper that has not been
bleached with chlorine.
Communicate with suppliers about
the need for totally recyclable or
reusable packaging materials.
With a careful examination of the cur
rent system, implementation of an edu
cation and recycling-based program,
hospitals can indeed reduce their waste
and save money at the same time.
Participating in waste reduction pro
grams will help your facility lead the
way in providing the best patient care
with concern for the safely' and well
being of your employees, patients, visi
tors and the communities you work in
and serve.
Notes
1.
Shaner, H. et al. (1993) An Ounce of
Pre vein ion- Waste Reduction Strategies for
Health Care Facilities. American Society
for Healthcare Environmental Services.
Chicago, IL.
HealthrCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.nohann.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
The rCF certification mark .mJ term
the sole property- of rhe Chlorine Free
Products Av4Xi.iti.-n mJ arc only med by authorued .mJ certified uxrs
. Red Bag
Reduction
Program
Adapted with permission from
Waste Reduction Remedies
by Stephanie C. Davis
1. Check Out Your Wastes
Has there been an analysis of disposal
costs by weight and volume to deter
mine a baseline?
Does someone routinely walk the
floors and review the trash scene by
department?
What is in the containers - and what
should be?
Are Regular Solid Waste containers
available wherever there are RMW
receptacles?
Are the Regular Solid and Red Bag
Waste containers easily accessible for
rhe staff that use them?
Is waste removal charged by depart
ment?
What changes can be made in the dis
posal contract?
2. Containers and Liners
Do Matter
What size containers are available, and
what sizes are really needed?
Are the current RMW containers stepons or open/lidless?
Can any newly purchased containers
be made out of rhe highest percentage
possible of recycled materials?
Are clear bags used for the Regular
Solid Wastes?
Are Red Bags cadmium and lead-free?
Is the ink used on Red Bags non-toxic?
3. Location, Location, Location
Where can one larger RMW container
be centrally located to replace numer
ous patient room containers? Utility
rooms? Drug dispensing rooms?
Specially constructed non-public area?
Has a spreadsheet inventory been
made of the type, number and depart
ment location of waste containers in
the facility?
Will reconfiguring the location of
step-on and open containers meet
facility needs?
Involve employees in container place
ment — they use the containers.
Remember, there may be some resist
ance to change, but patience, persever
ance and education go a long way.
4. Sharps Management
Are sharps disposed with the RMW?
Does a current contract or regulation
specify that Sharps and Red Bag Waste
be disposed of together, or is this con
vention?
Can a Sharps Container Reuse
Program be implemented with the
incumbent contractor or can it be
required on the next contract?
Who currently changes out the sharps
containers — facility staff or a con
tracted company?
Can sharps containers be made out of
the highest percentage possible of recy
cled materials?
5. Where are the Suction
Canisters Going?
Can the suction canisters be treated to
render them non-infectious and dis
posed of with the Regular Solid Waste?
Does a current contract or regulation
specify that Fluid Canisters and Red
Bag Waste be disposed together or is
this convention?
Can a Fluid Canister treatment pro
gram be introduced to the facility that
renders the contents non-infectious, is
cost-effective, is non-toxic, does not
impact the sewer system, and can be
implemented with relatively little effort
on the part of staff?
6. Efficient Pick-up Schedules
Do the internal disposal routes and
disposal schedules need adjustment or
a complete change?
Facility waste reduction practices and
changes impact staff and jobs. If a con
tracted housekeeping company is used,
are the employees on board with new
procedures?
Are there any related union issues that
warrant discussion?
7. Communication is Key
Are all regulated compliances in place?
Are signs and labels available in nonEnglish translations in addition to uni
versal symbols?
9. The P & 3Rs: Prevent,
Reduce, Reuse, Recycle
Continue to monitor, educate and
reduce wastes.
Talk to procurement administrators,
contractors and vendors about reduced
packaging requirements and “take
back” policies.
Learn from other hospital, industry and
government waste reduction programs.
Whenever possible phase-in re-usables
and “buy recycled" programs.
Start in-house material and chemical
exchange programs.
Start recycling programs for corrugated
paper, computer and mixed paper,
metal cans and glass bottles, and com
post food waste.
Do signs and labels match the color of
the type of wastes in the container to
which it refers?
Tie waste contracts to market prices
for recyclable commodities.
8. Educate All Employees
10. How Do You Define Success?
Knowledge helps with employee coop
eration. Cooperation helps reduce
waste and increase safety. Reduced
waste and increased safety reduces
costs.
The less red bag waste, the more solid
waste, the less solid waste, the more
recyclables, the less initially purchased,
the less total waste produced.
Education and re-education is an on
going process in health care facilities
impacted by constant change: staff, per
diems, students, patients and the tran
sient public.
Get employees engaged and thinking
about their waste habits by having
someone routinely walk the floors and
review the trash scene by department.
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green; 4 Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF ccrtiftcanon murk .ind term arc (he h*!c property of the Chlorine Free
Product* Avociaitan and are only used by autlk»n:cd and certified users-
Guidelines for
Optimizing
Waste
Segregation
By Hollie Shaner, RN, MSA
1. Hand washing sinks should have
waste containers lined with clear
bags beside them to capture paper
towel waste as solid waste rather
than red bags. Red bag waste is
automatically put into the biohaz
ard stream.
2. Every copier and printer should
have an appropriately sized recy
cling bin beside it, not a trash can.
Tremendous savings can occur once
this is implemented since paper
waste will be diverted from the
landfill, and directed to a recycling
facility where such wastes can actu
ally become revenue generators.
3. Every soiled utility area and every
department should have a “battery
waste” collection container. This
should be plainly labeled and readi
ly accessible so batteries can be
properly disposed of either as recy
clable or hazardous waste (depend
ing on the type of battery) and,
most importantly, kept out of the
‘incinerator-bound’ waste stream.
4. Clear bags should be used for solid
waste so staff can see through them
to know what they are handling.
80% of the waste cans should be
lined with clear bags, since MOST
of the waste will either be solid
waste or recyclable waste if properly
segregated.
5. Cadmium-free red bags should be
used to capture biohazard waste.
Red bags should be placed with
careful discretion and under the
control of the Waste Manager. Red
bags should not be randomly issued
for wastes. Housekeepers must
know specifically which containers
are supposed to be lined with red
bags. Other containers (the majori
ty of them) should be lined with
clear bags.
6. Cadmium-free sharps boxes and
containers should be used to cap
ture sharps waste. These contain
ers should be changed out by
housekeeping staff on a designated
schedule, and as needed to ensure
worker safety. All sharps contain
ers should be ordered under the
auspices of the Waste Manager to
avoid inadvertent procurement of
sharps boxes with cadmium for
colorant.
7. Wherever there is a vending
machine, there should be recycling
bins nearby to capture vending
machine-generated wastes. This
would include such wastes as alu
minum cans, #1 plastic soda bot
tles, newspapers, steel cans, etc.
8. Require vendors to take back pallets.
9. Flatten cardboard at the point of
generation. Transporting flattened
boxes enhances the efficiency of
transporting waste materials.
Flattened boxes reduce volume and
limit the amount of wasted airspace
in collection carts. This measure
can reduce the number of trips nec
essary to move materials to the
baler. Use a knife/blade to flatten
boxes quickly.
10. Rinse cans whenever possible. This
reduces odors and reduces the like
lihood of having insects swarming
around can collection containers. This
is especially necessary in food service
operations where the can waste is not
removed from the area daily.
1 1 .Pay attention to collection container
sizes and frequency of pick up. A
bag of segregated materials can
weigh much more than a bag of
mixed trash. Adjust container size
and collection frequency to achieve
rhe optimal situation of just enough
pick ups that are not too heavy to
retrieve. (A 40 pound waste han
dling weight limit for the house
keeping department is suggested to
prevent injury).
12. Institute mandatory facility-wide
waste education annually; post
waste program guidelines in every
department, on-line, and in
employee handbook; print recycling
guidelines on mugs, napkins, table
tents, in paycheck stuffers, newslet
ters, etc.
Reach for
Unbleached
Choosing
Chlorine-Free
Paper and Paper
Products
Of a hospital’s solid waste stream,
about 45% is paper and paperboard.1
Most hospitals have not made a conscions choice to move away from chlo
rinated paper products, resulting in the
continued purchase of office paper,
paper towels, bathroom tissue paper
and napkins that use chlorine in the
bleaching process.
When chlorine is utilized in the •
process of bleaching paper, high levels
of halogenated organic pollutants and
chlorinated compounds are released
into the environment, notably dioxins
and furans. Many of these chlorinated
pollutants are shown to cause numer
ous health problems including many
different kinds of cancer, reproductive
disorders, genetic damage and immune
system suppression. An average North
American pulp mill using chlorine
chemistry will use around 35-45,000
gallons of water per ton of pulp. A
chlorine free pulp mill will use 2,500 3,000 gallons of water per ton of pulp.
The math is simple — less water, less
pollutants, less energy, better for the
environment, better for human health,
better for industry.2
Hospitals should work with their
Purchasing/Materials Management
Department to choose paper that is
both recycled and chlorine-free, if pos
sible. Another area in which hospitals
can address this issue is in the pur
chase of paper products such as bath
room tissue paper, napkins, and perfo
rated, fold and roll towels. An effort
should be made to purchase recycled,
non-chlorinated versions of these prod
ucts. The Chlorine-Free Products
Association continuously updates a list
of chlorine-free paper suppliers and
producers of chlorine-free paper prod
ucts available on their website at
www.chlorinefreeproducts.org.
Purchasing/Materials Management
staff should ask their paper suppliers if
their paper is:
Processed Chlorine Free (PCF)
The Processed Chlorine Free label’ is
reserved for recycled content paper.
This includes all recycled fibers used as
a feedstock that meet EPA guidelines
for recycled or post-consumer content.
PCF papers have not been rebleached
with chlorine containing
compounds. Minimum of
30% post-consumer content is required.
Totally Chlorine Free (TCF)The Totally Chlorine Free label' is
reserved for virgin fiber papers. TCF
papers do not use pulp pro
duced with chlorine or
chlorine containing compounds as bleaching agents.
Elemental Chlorine Free (ECF)Elemental Chlorine Free paper DOES
NOT eliminate chlorine from the
bleaching process, it just uses another
form of the element, chlorine dioxide.
ECF pulp production continues to
result in the release of high levels of
halogenated organic pollutants and
chlorinated compounds into the envi
ronment.
Notes
I.
First, Do No Hann. Environmental Working
Group, 1997.
2.
w\vw.chlonnefrecproducts.org, Chlorine Free
Products Association, 2000.
3.
The PCF certification mark and term are
the sole property of the Chlorine Free
Products Association and are only used by
authorized and certified users.
Disposables
and Their
Alternatives
Item
Alternative
Underpads/chux
Reusable underpads; if adequate-sized
reusable underpads are used there is
rhe additional benefit of being able to
eliminate the drawsheet
Eggcrate mattresses
Purchase mattresses with built-in
eggcrates; minimize use of disposable
eggcrates
Single-use disposable Ambu bags
Reusable Ambu bags; can be used for
up to 8 years; cost more up front, have
reprocessing cost, but don’t become
waste for a long time
Single-use disposable ventilator circuits
Reusable ventilator circuits
Single-use disposable gowns
Reusable cloth gowns
Single-use dishware
Reusable dishware, both crockery and
cutlery
Single-sided copy machines for paper copies
Double-sided copiers, saves on paper
used, saves on waste generated
Single-use disposable pulse oximetry probes
Reusable pulse oximetry probes
Disposable diapers (for young and old)
Reusable diapers
Sharps containers
Reusable sharps containers
Single-use cardboard packaging
Reusable tubs for packaging regulated
medical waste
Single-use envelopes
Reusable inter-office mailers
Single-use disposable pillows
Reusable pillows
Single-use disposable bedpans
Reusable plastic or steel bedpans, or
dissolvable paper bedpans
Single-use urinals
Reusable plastic, steel or dissolvable
urinals
Single-use emesis basins
Reusable plastic or steel emesis basins
Single-use wash basins
Reusable plastic or steel wash basins
Single-use bowls
Reusable plastic or steel bowls
Single-use anti-embolytic products
Reusable anti-embolytic products
Single-use alkaline batteries
Rechargeable batteries
Disposable wash cloths
Reusable wash cloths
Disposable pitchers and cups
Reusable pitchers and cups
Pub 4 06 This publication is part of Going Given: A Resource Kit for Pollution Prevention in Health
lo r additional copies of this or other publications included in the kit, or to find out how to get a
complete kit, visit Health Care Without Harm on the Web at www.noharm.org.
This version: October 15, 2001
Recycling
Fact Sheet
Hospitals generate a tremendous
amount of trash and end up throwing
away valuable resources.
Comprehensive recycling and waste
minimization programs can save a
health care organzation both environ
mental and financial resources.
Facilities all across the country have
discovered that recycling programs can
simultaneously reduce disposal costs
and raise staff morale. Waste reduction
strategies go beyond recycling and
should emphasize waste minimization,
but recycling and reuse programs are a
critical aspect of any waste manage
ment and minimization program. As
community health providers, hospitals
should be pioneers in these important
environmental programs.
Below is a list of materials that should
be recycled in your facility:
In addition to recycling and reuse pro
grams, hospitals need to focus on cre
ating less toxic waste in the first place.
For example, hospitals need to adjust
their purchasing practices to favor
recycled content. Not only does this
help reduce the amount of pollution
generated to create these products, but
buying recycled also helps to stimulate
the market for the hospital’s recycled
materials.
a
Only 15% of the hospital waste stream
is classified ‘regulated’ or ‘potentially
infectious’, and must be handled as
such. The majority of hospital waste is
similar to that found in an office build
ing or hotel—mostly paper, cardboard,
metal and food waste. Much of this
waste can be diverted from landfills
and can reduce waste disposal costs
through the implementation of an
aggressive recycling program.
D
a
D
a
a
a
a
a
a
a
a
a
a
°
a
a
a
a
a
a
□
a
□
a
Batteries
° Ni-Cad
° Lead Acid
° Alkaline
° Mercuric Oxide
° Lithium
° Zinc Air
° Dry Cell
° Others
White Office Paper
Mixed Office Paper
Corrugated Cardboard
Aluminum
Glass
Newspaper
Magazines
Boxboard
Junk Mail
Books
Steel Cans
Silver
Toner Cartridges
Xylene
Fluorescent Lights
Formalin
Overhead Transparency Film
#1 PETE
#2 HDPE
#3 PVC
#4 LPDE
#5 PP
#6 PS
#7 Mixed
Implementing a hospital-wide man
datory paper recycling policy is a
necessity. Hospitals can see substantial
savings by diverting paper waste from
the landfill, and can actually generate
money from recyclers. Virtually all
waste haulers have some capacity to
collect recycled paper, while the few
that don’t can likely refer you to a
recycler in the area.
Easy steps to begin a recycling program
include:
E Even' copier and printer should
have a recycling bin placed beside
it, labeled RECYCLED PAPER in
large letters. There should not be
trash cans nearby, bur rather, kept
where other types of waste are usually generated.
°
Purchasing departments should
order paper with a high percentage
of recycled content.
0
Departments should make double'
sided copies where possible.
c
Paper can be reused in a plain
paper fax machine.
c
products that AMC can use in its labs.
The distillery is expected to reduce
AMC’s hazardous chemical waste pro
duction from 29 tons to 6 tons and
save $250,000 per year in disposal and
chemical purchasing costs.
Recycling Facts
n
In a lifetime, the average American
will throw away 600 times his or
her adult weight in garbage. This
means that each adult will leave a
legacy of 90,000 lbs. of trash for his
or her children.
u
The five primary material indus
tries—paper, steel, aluminum, plas
tics, and container glass—account
for 31 percent of U.S. manufactur
ing energy use.
n
You can make 20 cans out of recy
cled material with the same
amount of energy it takes to make
one new one.
n
Enough energy is saved by recycling
one aluminum can to run a TV set
for three hours or to light one 100
watt bulb for 20 hours.
n
In this decade, it is projected that
Americans will throw away over 1
million tons of aluminum cans and
foil, more than 11 million tons of
glass bottles and jars, over 4 and a
half million tons of office paper and
nearly 10 million tons of newspaper.
Almost all of this material could be
recycled.
Q
Incinerating 10,000 tons of waste
creates one job, landfilling the same
amount creates 6 jobs, recycling the
same 10,000 tons creates 36 jobs.
■
Every' Sunday, the United States
wastes nearly 90% of the recyclable
newspapers. This wastes about
500,000 trees.
Substitute reusable inter-office
mailers in place of single-use
envelopes.
It is important to remember that each
area in a hospital has special needs and
should be treated as an independent
system. An Ounce of Prevention: Waste
Reduction Strategies for Health Care
Facilities (available through the
American Hospital Association) is an
excellent resource on how to imple
ment a recycling program in your hos
pital, and comprehensively addresses
the departmental concerns that need
to be taken into consideration.
A Case Study
Albany Medical Center (AMC), a 500bed research hospital in upstate New
York has a model recycling program.
The program recycled 16 million
pounds of waste and saved rhe hospital
$4 million in its first six years. The
facility’ is now recycling 43 percent of
its total waste stream. In addition to
the host of typical items it recycles,
such as paper, cardboard and steel
cans, AMC is able to recycle five dif
ferent types of waste chemicals into
usable products through rhe use of a
$75,000 chemical distillery it built in
1995. The distillation center can con
vert waste alcohol, formalin, xylene,
mineral spirits and paint into pure
■
One tree can filter up to 60 pounds
of pollutants from the air each year.
Health rCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax; 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
ins wit*
INK
•-•>13
Tbe PCF certification mark and term are the a-Iv prvpcnv of rhe Chk»nr.e Free
Products Association .mJ arc only used b> authorized .mJ certified users.
Waste
Minimization
[Resources
Air Force Environmental Exchange.
PRO-ACT Fact Sheet: Management of
Medical/Infectious Waste. Environmental
Quality Directorate HQ Air Force
Center for Environmental Excellence,
October, 1998.
Citizens’ Environmental Coalition
(CEC). Environmentally Safe Hospitals,
Reducing Waste and Saving Money. A
Resource Guide for New York City
Hospital Materials and Waste Managers
New York: CEC, 1999. 518-462-5527
Barlow, Rick.“Medical Waste Becomes
Monster in Cost-Cutting Fight.”
Hospital Material Management
December, 1991:1-10.
City of Palo Alto Regional Water
Quality Control Plant. Best Management
Practices for Hospitals and Medical
Facilities. Palo Alto Regional Water
Quality' Control Plant, 2501
Embarcadero Way, Palo Alto, CA
94303; (415) 329-2598
Bisson, Connie, Glenn McRae and Hollie
Shaner, RN. An Ounce of Prevention:
Waste Reduction Strategies for Health
Care Facilities. Chicago: American
Society for Healthcare Environmental
Services, 1993. Available from the
American Hospital Association.
AHA Publication # 057007.
1-800-AHA-2626
Brady, Lorraine. “Start-up Establishing
Infectious Medical Waste Disposal
System." Health Industry Today
August, 1994:1-14.
Boston University Corporate Education
Center. A New Prescription: Pollution
Prevention Strategies for the Health Care
Industry. Proc, of a Workshop of the
Boston University Corporate Education
Center, Oct. 1998, Tyngsborough,
Massachusetts.
Brown, Janet. Guide to Waste
Management. New York: Beth Israel
Medical Center, 1997. 212-420-2442
Byrns, George and Thomas Burke.
“Medical Waste Management
Implications for Small Medical
Facilities.” Journal of Environmental
Health Vol. 55, No. 3. Nov/Dcc.
1992:12-15.
California Integrated Waste
Management Board (CAL EPA).
Online. Internet. October, 2001.
Available FTP: www.ciwmb.ca.gov/
BizWaste/Factsheets/Hospital.htm
r-»
O
O
CXJ
*
OJ
.n
o
c
o
Z
Canadian Centre for Pollution
Prevention and Broadhurst
Environmental Management Inc.
Health Care Pollution Prevention and
Environmental Management Resource
Guide. 1-800-667-9790
City of Palo Alto Regional Water
Quality Control Plant. Pollution
Prevention for Hospitals and Medical
Facilities. Palo Alto Regional Water
Quality' Control Plant, 2501
Embarcadero Way, Palo Alto, CA
94303; (415) 329-2598
Davis, Stephanie. “Ten Steps To
Consider Towards Implementing A Red
Bag Reduction Program.” Waste
Reduction Remedies. 2000.
Department of Toxic Substances
Control, Office of Pollution Prevention
and Technology Development. Western
Regional Pollution Prevention Network
(P2 West). Online. Internet. (2000)
Available FTP: www.westp2net.org/
sector/healthcare.htm
Department of Veterans Affairs. VHA
Program Guide 1850.1 Recycling Program.
Washington, DC: Environmental
Management Programs Office, Veterans
Health Administration, 1998.
Environmental Working Group and
Health Care Without Harm. First, Do
No Harm. Washington, DC: 1997.
Available FTP: www.ewg.org/pub/
home/HCWC/hcwh.html
Garvin, Michael L. “Reducing Waste
Volumes: 3 Obstacles to Overcome.”
Health Facilities Management
June, 1990:32-42.
Goldberg, Michael E. et al. “Medical
Waste in the Environment: Do
Anesthesia Personnel Have A Role to
Play?” Journal of Clinical Anesthesia
Vol. 8:1996.
Kentucky Pollution Prevention Center
(KPPC). Managing Your Medwaste for a
Healthier Bottom Line. (1996). Kentucky
Pollution Prevention Center
420 Lutz Hall, Louisville, Kentucky
40292; (502) 852-0965 Available FTP.
www.kppc.org/Publications,/Videos/
medwastevideo.cfrn
Kerley, Frank R. and Brent E. Nissly.
“Total Quality Management and
Statistical Quality Control: Practical
Applications to Waste Stream
Management.” Hospital Material
Management Quarterly Vol. 14, No. 2.
Nov. 1992:40-59.
Nelson, Julie A. and Larry A. Gibson.
Pollution Prevention Wbrks for Iowa:
Health Care Case Summaries. (January,
1996). Iowa Waste Reduction
Assistance Program, Waste
Management Assistance Division, Iowa
Department of Natural Resources, 900
East Grand Avenue, Des Moines, IA
50319-0034; (515) 281-8927
New York State Department of
Environmental Conservation.
Environmental Self-Assessment for
Health Care Facilities: A Quick and
Easy Checklist of Pollution Prevention
Measures for Health Care Facilities.
Online. Internet. February', 2000.
Available FTP: www.dec.state.ny.us.
1-800-462-6553
Riggle, David “Solid Waste Surgery:
Advance Hospital Recycling.” BioCycle
February, 1994:34-37.
Rau, Edward, et al. “Minimization and
Management of Wastes from Biomedical
Research.” Environmental Health
Perspectives Vol. 108, Suppl. 6.
Dec. 2000:953-77.
Ridley, Keith. Writing a Waste Reduction
Plan for Healthcare Organizations. Center
for Industrial Services, University' of TN,
1997. 615-532-4926
Shaner, Hollie. “Pollution Prevention for
Nurses: Minimizing the Adverse
Environmental Impacts of Health Care
Delivery’." Vermont Registered Nurse
Vol.62, No. 4. 1996:1-2, 8-9.
Shaner, Flollie and Glenn McRae. The
Guidebook for Hospital Waste
Reduction Planning and Program
Implementation. Chicago: American
Society for Healthcare Environmental
Services, 1996. Available from the
American Hospital Association.
AHA Publication # 057037
1-800-AH A-2626
Shaner, Hollie and Glenn McRae.
“Invisible Costs, Visible Savings:
Innovations in Waste Management for
Hospitals.” Surgical Services
Management Vol. 2, No. 4. April 1996.
Shaner, Hollie and Glenn McRae. No
Time to Waste, Resource Conservation for
Hospitals. Professional Development
Scries of the American Society for
Healthcare Environmental Services.
Chicago: American Society' for
Healthcare Environmental Services,
1997.
Shaner, Flollie and Glenn McRae.
Eleven Recommendations for Improving
Medical Waste Management. Nightingale
Institute for Health and the
Environment. Online. Internet.
December 1997.
Available FTP: www.nihe.org
Wagner, Kathryn. Environmental
Management in Healthcare Facilities.
Philadelphia: W.B. Saunders Company,
1998.
Waste Reduction and Disposal Options for
Specific Hospital Wastes. NC Division of
Pollution Prevention and Environmental
Assistance and NC Division of Waste
Management. Online. Internet. August,
1996. Available FTP: www.p2pays.org/
ref/01700239.pdf
U.S. Environmental Protection Agency.
Project Summary: Hospital Pollution
Prevention Case Study. Washington:
August, 1991. EPA/600/S2-91/024
U.S. Environmental Protection Agency.
Guides to Pollution Prevention: Selected
Hospital Waste Streams. Washington:
June, 1990. EPA/625/7-90/009
HealthrCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www. noharm .org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
SOYINK
The PCF ccrtihcatkin mark and term arc the xlc property or the Chlorine Free
Products Assxiinun and are unh used by authorized and certified user*.
Environmentally
Preferable
Purchasing
How-To Guide
What is environmentally
preferable purchasing?
Why is the purchasing
stage so important?
Environmentally preferable purchasing
(EPP) is the act of purchasing products/services whose environmental
impacts have been considered and
found to be less damaging to the envi
ronment and human health when
compared to competing products/services. EPP also includes the gradual
and ongoing process in which a hospi
tal continually refines and expands the
scope of its efforts to select environ
mentally sound, healthy and safe prod
ucts and services. A hospital’s choice
to implement EPP is an important part
of a larger system of a hospital’s prac
tices that support the integrity of both
business and environmental decisions.
EPP may be as simple as buying recy
cled paper or as complex as considering
the environmental impact of a product
at each stage of its life, from when it is
manufactured to when it is disposed of
as waste.
Purchasing departments are the central
control point for nearly every product
or service procured by the hospital.
This is where the money is transferred
from hospital to vendor and where
contracts are developed. It is at this
stage that leverage can best be applied
to the vendors, making it an effective
place to implement actions that reduce
environmental impact.
What are the benefits
of EPP?
By carefully selecting goods and servic
es, hospitals can:
a reduce costs due to lower overhead,
avoid waste disposal, liability or
occupational health costs
c
Why is it less costly to
make improvements at
the point of purchase?
Correcting a problem close to its
source is less costly than taking action
downstream. Downstream corrections
require a greater degree of technical
complexity' and labor to correct and
often result in adverse publicity. A
hospital that tries to save money by
overlooking the environmental aspects
of a product during the purchasing
stage is likely to incur much greater
expenses later on.
How Costs Increase the Further
Downstream a Problem is Addressed
take advantage of positive publicity
and promotion potential
D
significantly improve their impact
on the overall quality of the envi
ronment
n
provide a healthier environment for
patients, workers and employees
through reduced exposure to clean
ers, solvents, paints, and other haz
ardous materials.
Additional Cost to Buy an
Aneroid (instead of Mercury)
Sphygmomanometer
Clean up Costs for Broken
Mercury Sphygmomanometer
$1009
Cost of Incinerator
Environmenatal Control to
Remove Mercuiy Normalized
for one Mercury
Sphygmomanometer
Reference: Mercury Elimination and Reduction
Challenge (MERC), “Mercury in the Health Care
Sector: The Cost of Alternative Products,"
November, 1996, pp 1424
Flowchart from the
Hospitals for a Healthy Environment
Environmentally Preferable Purchasing
"How To" Guide
Step 1. Establish a
Multidisciplinary team for EPP
V
Setting up the
Environmentally
Preferable Purchasing
(EPP) team
An EPP team is comprised of hospital
professionals from different areas work
ing together to foster a new purchasing
culture. This team should coordinate
its activities with the facility-wide
environmental team and the product
review committee (s). The leader of
the team should be someone whose
administrative responsibilities include
ensuring that the EPP Project is fully
implemented.
Why is an EPP
team necessary?
UJ
Pilot test successful
I
The diverse perspectives of members
from various departments can chal
lenge current practices and promote
innovative solutions. A team can work
together to create pilot projects and
provide effective solutions to obstacles.
If each department is part of the
process, there will be greater buy-in to
changes in practices and products. A
dedicated team can also motivate the
purchasing and other departments to
implement environmentally preferable
purchasing. The facility-wide environ
mental team is looking at the whole
picture, and may not have the
resources to implement environmental
ly preferable purchasing without the
assistance of a dedicated EPP team.
Some hospitals may find that the prod-a.
uct review committee or the facilitywide environmental team are suffi
ciently interested in EPP that a
separate EPP team is not necessary.
The team should include:
n
representation from all relevant
departments
n
someone with management respon
sibility
u
people with a passion for and
understanding of the ecological
focus of the team
Membership can include
representation from:
□
Central Services
□
Clinical Staff
a
Communication/Public Relations
□
Environmental (Ecology) Team
□
Environmental Services
0
Facilities Operations (physical
plant, operations, logistics, and
security)
n
Financial Services (Accounting)
a
Food Services
a
Group Purchasing Organization
(GPO)
a
Infection Control
■
Laboratory services
a
b
Materials Management (purchasing, contracting and distribution
services)
a
Prime Distributor
°
Risk/Safety Management
n
Waste Management /
Housekeeping
n
n
Review pressing environmental con'^Berns of the hospital and available
resources so that the committee can
be informed when deciding on goals.
3. Decide on environmentally prefer
able purchasing goals that are specific,
measurable, and can be completed in a
specific time period. For example:
a
Increase purchase of recyclables
or reusables by 30% by the next
fiscal year.
■
Reduce packaging waste or total
solid waste by 20% in 12 months.
0
Reduce energy or water use by
10% every six months for 5 years.
4. Develop an implementation timeline.
a
The timeline should be realistic
and allow time for research and
evaluation of alternatives, educa
tion of affected parties, and contin
uous evaluation of pilot.
n
Be creative when deciding on
method to achieve goals. Reducing
hazardous waste from the histology
lab could involve changes in prac
tice (not using more solvent than
necessary), capital equipment
expenditures (buying an autoana
lyzer that uses microamounts), or
procedure (switching to a less toxic
fixative). Involve the workers from
that department in soliciting ideas
for how to meet the goal.
n
Continuous evaluation should be
part of any EPP program. Set in
place mechanisms for obtaining
continuous feedback from employ
ees and product users, evaluating
that feedback and using it to
improve the program or a specific
product.
°
Create a tracking system.
Reduce purchase of mercurycontaining products by 80% by
next year.
Actions to implement
environmentally
preferable purchasing
1. Request support for EPP goals from
top management in the form of a poli
cy statement, RFP language, job
descriptions, or other support.
2. Develop policies and procedures to
ensure the implementation of the
environmentally preferable purchasing
practices:
■
Determine in writing who is
responsible for ensuring that poli
cies are followed and how they will
be held responsible (for instance,
through periodic reporting).
u
Develop an audit process so that
performance is periodically
reviewed. The audit process should
incorporate a system for the cele
bration and duplication of success
es, and the recognition and rectifi
cation of projects or products that
did not work.
Determining goals
and objectives of the
EPP team
1. Consult with facility environmental
team to determine which EPP goals
might fulfill the main environmental
goals of the institution.
Reduce purchase of products that
become hazardous waste by 10% in
the next contract.
5. Determine educational needs to
implement EPP Education is a critical
part of implementation. The EPP team
should consult with the inservice
training department to discuss educa
tional needs, such as education of:
a
purchasers and users on the need
for EPP;
a
3. Using the measurable goals deter
mined above, choose a small, manage
able pilot project. For example:
top management on what support
is needed to implement EPP;
□
Replace mercury sphygmo
manometers with aneroid equip
ment in one department.
how new products/practices will be
evaluated and what feedback is
desired;
□
how employees are to use the new
product;
a
other affected parties;
o
new employees at orientation; and
a
vendors, manufacturers,
distributors, and GPO.
a
□
□
□
Determine in writing who is
responsible for the audit process.
Work with histology lab to find
mercury-free replacement for a spe
cific reagent in a specific process.
Include environmental criteria,
such as battery recycling or energy
efficiency, in next major equip
ment or service solicitation.
Implementation of
specific goal/pilot
project:
1. Implementation:
c
It goal involves replacement or
focus on specific product, work
with product selection committee
or standardization committee in
hospital and GPO to determine
process (tor instance, writing envi
ronmental specifications for RFP).
c
Determine and publicize timeline
for implementation of specific goal.
n
Determine who is responsible for
ensuring timeline and goals are met.
c
Determine educational needs to
implement EPP project. Create a
written plan for education of affect'
ed parties regarding implementa
tion of this particular project,
including who is responsible for the
education.
n
Implement purchase.
2. Continual Improvement:
a
Determine if measurable goal was
met.
D
Request feedback from affected
parties.
c
Review process.
G
Incorporate feedback into action
plan for next project or improve
ment of this one.
c
Keep records and track progress.
3. If Goal Was Successfully Met:
c
Publicize success to hospital and
wider community.
D
Assess possibility of expansion of
pilot project or determine next spe
cific goal.
c
D
To determine next specific project,
consider introducing additional
environmental considerations,
raising the measurable goal, or
expanding the program.
Track and report on progress.
4. If Goal Was Not Met:
a
Do not be discouraged!
a
Determine the causes of not meet
ing the goal.
a
Brainstorm on how to correct the
shortcoming, move forward and be
creative!
n
Choose an interim goal or pilot
project to implement to get back
on track.
n
Move forward on the new goal or
pilot project.
Additional resources
Lists of resources, a detailed EPP edu
cation matrix, flow chart of actions,
links to specifications, discussion of
related issues, suggestions for trou
bleshooting, and more details on EPP
are available at http://h2e.ashes.org.
This is a product of the Environmentally
Preferable Purchasing workgroup of Hospitals for
Healthy Environment, a cooperative project
between the US EPA and the American Hospital
Association.- Lara Sutherland (Massachusetts
Office of Technical Assistance), Christopher
Kent (US EPA), Catherine Galligan (University
of Massachusetts-Lowell Sustainable Hospitals
Project), Tim Washburn (Catholic Healthcare
West), Kinley Deller (King County Solid Waste),
Patrick Eagan (University of WisconsinMadison), Timonie Hood (US EPA Region 9),
Glen Macri (Becton Dickinson), Layne Nelson
(Minnesota Department of Administration),
Russ Sylvester (Premier, Inc.), Joan Roberts
(Novation), John Mateka (Memorial Regional
Hospital), Sidney Pittman (Halifax Medical
Center), Wayne Warren (Veterans
Administration).
Health rCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: 4 Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
INK
rhe PCF eradication nurk .mJ term are the m?Ic property ot the Chlorine Free
Product* A'Mxbtmn .mJ arc only uxd by authoeixd .mJ certified uxrrs
Sample letter
to Group
Purchasing
Organizations
This is modeled on a letter
sent to GPOs by the Michigan
Hospital Association.
Fred Representative
Group Purchasing Organization
5 GPO Drive
Anywhere, PA
Dear Mr. Representative:
In 1998, the American Hospital Association and the United States Environmental
Protection Agency signed a Memorandum of Understanding setting aggressive
goals for the health care industry to minimize the volume and toxicity of medical
waste. [Your organization] has established a Task Force on Hospitals for a Healthy
Environment [or other efforts] to coordinate these efforts in [your areal.
Many organizations dedicated to environmental protection are closely examining
health care operations, and along with the communities we serve, are asking if
health care as an industry is doing all that it can to protect and preserve the natural environment. In concert with the AHA and the USEPA, [your organization]
calls on manufacturers, suppliers and group purchasing organizations to begin
work toward implementation of the following initiatives:
c
Eliminate Mercury: As an industry, we must identify and develop alternatives
to mercury and mercuric compounds. Health care is identified as a major con
tributor to mercury in the environment and we must do all we can to remove
this bio-accumulative toxin from our operations. Even trace amounts in com
mon health care products should be identified and eliminated.
Eliminate the use of PVC plastics: The manufacturing and incineration of
PVC plastics used in the health care industry have been identified as a major
source of dioxin in the natural environment. It is critical that the health care
industry identify and implement competitively priced alternatives to PVC.
a
Reduce Wasteful Packaging: Source reduction must be a major focus to enable
the health care industry to achieve waste reduction goals. The elimination of
unnecessary packaging, return programs, elimination of polystyrene foam as pack
aging material and other waste reduction efforts will prove critical to success.
n
Recycled Content: Successful waste recovery' and recycling programs will be
essential to meeting waste reduction goals. Recycling programs depend upon
strong markets for recovered materials. As an industry', recycled content
should be included in as many products as possible to provide demand for
recovered materials. Additionally, efforts should be made to replace hard-torecycle plastics with commonly recycled materials.
[Your organization] is committed to fostering healthy communities and environ
mental safety and health. We recognize that the health and well-being of the
people and communities we serve is fundamentally connected to the health and
vitality of the environment and natural world we all share, and we look forward
to working with all segments of our industry to position health care as a leader in
building sustainable communities.
For more information, about these efforts, please contact [your organization].
Sincerely,
in
[Representative]
[Your Organzation]
Latex*
Allergy in
KsEVib Care
Fact Sheet
* natural rubber latex
Latex AllergySymptoms and Causes
Since 1987, when the Centers for
Disease Control and Prevention
(CDC) recommended universal pre
cautions, the increased use of natural
rubber latex gloves in health care set
tings has been associated with an
increase in reported natural rubber
latex allergies among both patients and
workers. Prevalence studies indicate
that 6-17% of the exposed health care
workforce has become allergic to
latex.1 Symptoms range from irritating
to life-threatening.
Ten years later, in June 1997, the
National Institute for Occupational
Safety and Health (NIOSH) published
an Alert - “Preventing Allergic
Reactions to Natural Rubber Latex in
the Workplace” — which, among other
recommendations, called for education
to inform workers of the symptoms of
latex allergy. These symptoms include
dermatitis, urticaria, rhinitis, nasal, eye
or sinus symptoms, asthma, and ana
phylaxis.2 Deaths have been reported
as well.' Latex is recognized by
NIOSH as a hazard to the health of
exposed workers.
Latex allergy has become an increas
ingly serious threat to health care
workers (housekeepers, lab workers,
dentists, nurses and physicians) who
experience frequent or prolonged
exposure to natural rubber latex
through inhalation and exposure to
mucous membrane or disrupted skin.
Sensitization occurs through contact
with latex proteins. Powder on gloves
is a vehicle for sensitization. Powder
increases the probability of sensitiza
tion as it allows direct contact of
aerosolized latex proteins with
mucous membranes of the eyes and
respiratory tract.
For many allergic workers the common
denominator is, “I have been using
latex gloves for years, why is this a
problem now?” The number of expo
sures necessary for sensitization varies
depending on the individual. A health
care worker can use latex gloves for
many years before developing a latex
allergy.
For most sensitized people, the symp
toms of skin rashes, runny nose, and
itchy eyes persist for a very long time.
For others, the rashes and runny nose
quickly become breathing problems
such as asthma, airway obstruction, and
extreme spasms in the throat (laryngospasm). For still others, the first
symptom may be life-threatening shock
(anaphylaxis). No immunotherapy or
desensitization exists for latex allergy.
Each systemic reaction comes with less
provocation; each reaction is worse.
For a summan’ of the reactions associ
ated with latex gloves, see Table 1.
Patients with spina bifida, and patients
with congenital genitourinary abnor
malities who are heavily exposed to
natural rubber latex through surgical
procedures and contact with latex
catheters show sensitization rates as
high as 18-73%.4,5 Patients who have
undergone as few as three surgical pro
cedures may be at a higher risk of
developing latex allergy.
Not only direct contact with latex, but
also exposure to the airborne latex pro
teins carried on powder can sensitize
an individual and elicit an immune
response (allergic reaction). Therefore,
only avoidance of exposure to latex
material and aeroallergens will prevent
latex allergy from developing in work
ers and patients.6
r
e e
Cause of Reaction
Terms Used or Description
Signs and Symptoms
Cause(s)
Irritant contact
dermatitis
Irritation
(non-allergic irritation)
Dry, crusty, hard bumps, sores,
and horizontal cracks on skin may
manifest as itchy dermatitis on
the back of hands under the
gloves
Direct skin irritation by
gloves, powder, soap/detergent, scrubs, and/or incom
plete hand rinsing and dry
ing
Allergic contact
dermatitis
Type IV delayed
hypersensitivity
Red, raised, palpable area with
bumps, sores, and horizontal
cracks may extend up the forearm.
Occurs after a sensitization peri
od. Appears several hours after
glove contact and may persist
many days.
Exposure to chemicals used
in latex manufacturing,
including accelerators, bio
cides, antioxidants (e.g.,
thiurams, carbamates, and
benziothiazoles)
Wheal and flare response or itchy
redness on the skin under the
glove. Occurs within minutes,
fades away rapidly after removing
the glove. In chronic form may
mimic irritant and allergic contact
dermatitis. Symptoms can include
facial swelling, rhinitis, eye symp
toms, generalized urticaria, respi
ratory distress, and asthma. In
rare cases, anaphylactic shock may
occur.
Exposure to proteins in
latex on glove surface
and/or bound to powder
and suspended in the air,
settled on objects, or trans
ferred by touch.
T H
C A
RE
FACT
Table 1. Types of reactions associated with latex gloves
Allergy to latex
proteins
Type I hypersensitivity
IgE/histamine mediated
reaction
TEX
ALLERGY
IN
H E A
L
Allergy contact sensitivity
<
Source: American Nurses Association. Latex Allergy: Protect Yourself, Protect Your Patients (brochure). Washington, D.C. ANA, 1996.
Worker Protection
According the Occupational Safety
and Health Law of 1970, employers
have a responsibility to provide a
workplace free from recognized haz
ards that are causing or are likely to
cause death or serious physical harm
to employees.
Recommendations
for a Latex Safe
Work Environment7
1. Use non-latex — and non-chlorine
(non-vinyl and non-neoprene)
containing’’ — examination gloves
in all health care settings.
2. Use latex-free equipment in resus
citation and invasive procedures.
3. Identify products that contain
latex, including surgical gloves and
other medical devices:
o
locate non-latex alternatives
and
°
plan, evaluate and implement
the use of non-latex alterna
tives.
4. Provide education for nurses and
other health care workers to ensure
an understanding of latex allergy,
including:
°
°
°
routes of exposure, sensitization
and reactions;
procedures for reporting acute
and chronic occupational ill
ness;
protocols for treatment and
accommodation of sensitized
workers;
°
submit written reports (retain
ing copies) of their symptoms to
their supervisors and the occu
pational health department
(when available); and
°
report adverse health effects
resulting from the use of latex
gloves and other latex medical
devices to the FDA MedWatch
Program: tel: 1-800-FDA-1088
or fax: 1-800-FDA-0178.
5. Provide education for nurses and
physicians to:
°
recognize signs and symptoms
of latex allergy in patients;
°
safely care for latex allergic
patients; and
°
learn treatment protocols for
patients with acute allergic
reactions to latex.
6. Identify health care providers with
expertise in treating latex allergy to
provide care for latex allergic nurs
es, other health care workers, and
patients.
Table 2. Non-latex and non-chlorine (non-vinyl and
non-neoprene) containing gloves
7. In some states it is rhe law to
report cases of latex-induced occu
pational asthma to the Department
of Public Health, as in
Massachusetts, where all cases
must be reported to the
Department of Public Health,
Occupational Health Surveillance
Program (tel: 617-624-5637).
Non-latex Gloves with
Barrier Protection Equal
io or Better Than Latex
variety of non-latex gloves made of
alternative materials, with barrier pro
tection equal to or better than latex
gloves, are available (see Table 2).
The protective characteristics of each
material must be taken into considera
tion in relationship to the purpose for
which the glove will be used.
Information on how to select medical
gloves and a list of non-latex glove
alternatives are available from the
Sustainable Hospitals Project (SHP) at
the University of Massachusetts
Lowell. This information can be found
online at www.sustainablehospitals.org
or contact the SHP directly at 978934'3386 or shp@uml.edu.
■t summary', health care practitioners
and their employers must protect
themselves and others against latex
sensitization and allergy. Important
steps include:
■
Use non-latex and non-vinyl gloves
that offer barrier protection equal
to or better than natural rubber
latex.
■
Learn to recognize the signs and
symptoms of latex allergy in your
self, co-workers and patients.
Manufacturer
Glove
Ansell-Perry
800-321-9752
www.ansellhealthcare.com
Nitra-Tex™ nitrile exam glove
Nitra-Touch® nitrile exam glove
Elite™ polyurethane surgical glove
Best Manufacturing Co.
800-241-0323
www.bestglove.com
N-DEX® and Nitra-Care® gloves
ECI Medical Technologies,Canada
902-543-6665
www.ecimedical.com
Elastyren® family of synthetic copoly
mer medical gloves
•
Maxxim Medical
800-727-7951
www.maxximedical.com
SensiCare™ Nitrile exam glove
SensiCare™ NXP exam glove
SensiCare™ polyurethane exam glove
SensiCare™ polyisoprene surgical glove
Safeskin Corporation
800-462-9993
www.safeskin.com
Safeskin Blue Nitrile
Safeskin Purple Nitrile™
SmartCare Inc.
800-822-8956
www.smartcare.com
Nitra PF™
Tillotson Healthcare Corporation
800-445-6830
www.thcnet.com
Dual Advantage
Pure Advantage
Source: Sustainable Hospitals Project Clearinghouse, www.sustainablehospitals.org
If you or anyone has the signs and
symptoms of latex allergy:
□
report the signs and symptoms to
supervisors, managers, and occupa
tional health providers immediately;
a
inform all your healthcare
providers — physicians, dentists,
nurses — that you have latex aller
gy and that you must avoid expo
sure to all latex products including
latex gloves; and
s
wear a medical alert bracelet.
Only with increased awareness, educa
tion, reporting, and support will health
care practitioners be enabled to protect
themselves, their co-workers, and their
patients from sensitization and poten
tially life-threatening reactions to latex.
Health care practitioners and employ
ers will not be able to prevent them
selves, their employees, and patients
from sensitization and potentially life
threatening latex reactions unless latex
is removed from the workplace.
Increasing attention to latex allergy'
education and latex-safe protocols for
patient care is essential for a safe envi
ronment for workers and patients alike.
References
1 <S< 2. US Department ot Health and Human
Sen-ices. Public Health Sen-ice. Centers for
Disease Control and Prevention. NIOSH
Alert. Preventing allergic reaction to natural
rubber latex in the workplace. June, 1997;
NIOSH publication, pp 97-135.
3
US Department of Libor. Occupational
Safety and Health Administration.
Technical Information Bulletin: Potential for
Allergy to Natural Rubber Latex Gloves and
Other Natural Rubber Products.
Washington, DC: OSHA, April 12, 1999.
4
Meeropol, E., Kelleher R, Bell SI, &. Leger
R., 1990. Allergic reactions to rubber in
patients with myelodysplasia. New England
Journal of Medicine. 1990: 323:2072.
5.
Kelly, K., Pearson, M , &. Kurup, V A clus
ter of anaphylactic reactions in children
with spina bifida during general anesthesia.Epidemiologic features, risk factors, and
latex hypersensitivity.
6.
Poley, GE. Slater JE, Latex Allergy. J
Allergy Clin Immunol 2000:105(6)-10541062.
7
Source of recommendations: Massachusetts
Nurses Association, Latex Allergy Position
Statement, (1997).
8.
Polyvinyl chloride (also known as PVC or
"vinyl") and polychloroprene (“neoprene")
are the chlorine-containing materials used
to manufacture examination gloves.
Chlorinated materials are of concern
because they can contribute to dioxin emis
sions from incinerators.
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234-0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF certification nurk and term arc the sole property v( (he Chlonnc Free
Prixincts Auociation and arc only ux*J by authorked .mJ certified mere.
T@ Eliminate
Qiitaraldehyfe
Fact Sheet
A FACT SHEET OF THE
SUSTAINABLE HOSPITALS
PROJECT
SHP is a project within the University of
Massachusetts Loivell Center for
Sustainable Production, providing
technical support to health care.
ivww.sustainablehospitals.org
Hospital disinfection is serious busi
ness. When glutaraldehyde was first
marketed in the early 1960’s‘, it was
good news. Effective alternatives were
sought to the highly toxic, irritating
and carcinogenic disinfectant
formaldehyde. However, reports of
serious health effects from glutaralde
hyde exposure were published shortly
thereafter and ever since. Today, 40
years later, there are alternatives that
offer high level disinfection while pro
tecting health care workers and the
environment.
Reasons for Elimination
1. Glutaraldehyde (GA) is a potent
occupational skin irritant and sen
sitizer.1,4
2. Glutaraldehyde exposure in hospi
tals is a recognized cause of occupa
tional asthma510 in many industrial
ized nations (England, Australia
and others) although it is not regu
lated in the United States. Studies
demonstrate that adverse respirato
ry health effects may occur at levels
below 0.2 ppm, the current NIOSF1
Recommended Exposure Limit
(REL).11,12
3. Anecdotal reports suggest that GA
exposure has been associated with
the development of chemical sensi
tization disorders.15 This condition
results in an intolerance not only
to glutaraldehyde, a sensitizer, but
to many other classes of chemicals
as well.
4. Patients, visitors, and hospital staff
may be needlessly exposed to glu
taraldehyde vapors in patient rooms
and clinical areas where open bins
or poorly ventilated reprocessing
units are in use.
5. Alternatives to glutaraldehyde are
available that maintain infection
control standards14'17 and do not
cause undue wear and tear on sen
sitive medical devices.
6. Alternatives to glutaraldehyde are
available. These alternatives are
safer both for workers (the risk of
skin and respiratory sensitization is
avoided) and for the environment.
7. It’s smart to stay ahead of the
game. OSHA is currently develop
ing a Permissible Exposure Limit
(PEL) for glutaraldehyde.
Observers suggest that a 0.05 ppm
ceiling limit may result due to evi
dence that respiratory sensitization
can still occur at the NIOSH REL
of 2 ppm. Other countries have
lowered or are in the process of
lowering their “ceiling” limits to 0.1
ppm or 0.05 ppm. In the US, the
American Congress of Government
Industrial Hygienists (ACGIH)
recently lowered their Threshhold
Limit Value (TLV - 15 min STEL)
to 0.05 ppm.18
8. The alternatives will be cheaper in
the long run:
Direct costs of using glutaralde
hyde include: special ventilation
hoods, improved general ventila
tion, construction or purchase of
enclosed disinfection stations, per
sonal protective equipment, educa
tion and training programs, ongoing
monitoring programs, chemical
neutralization solutions, mainte
nance of a glutaraldehyde emer
gency spill team, and work practice
aids such as absorbent mats, pour
ing nozzles, etc.
Indirect costs — largely over
looked — include: employees with
occupational dermatitis, employees
with occupational asthma, lost
work time, workers’ compensation,
costs of replacement labor, costs of
managing staff, patient and com
munity' relations. Future costs may
include: compliance with a new
OSFIA PEL and action from local
POTWs (publicly operated treat
ment works) regarding the dumping
of aldehydes, such as glutaralde
hyde, down the drain.19,20
9. A plan to eliminate or phase-out
glutaraldehyde is consistent with a
public health approach:
PREVENTION. It makes sense to
eliminate highly toxic and sensitiz
ing substances from the hospital
environment when alternatives
exist that are feasible, effective
and sustainable.
10. Glutaraldehyde has successfully
been eliminated — or dramatically
reduced — in dozens of hospitals.
The success of these hospitals is the
best testimony for the benefits of
change.
For more information
Contact the Sustainable Hospitals
Project (SHP) by:
Phone (978) 934-3386
Email: shp@uml.edu
Mail: Sustainable Hospitals Project,
Kitson 200, One University Avenue,
Lowell, MA 01854.
Visit the SHP website for information
on alternative products and practices:
www.sustainablehospitals.org
Notes
1.
2.
3.
Stonehill AA, Drop S Borick PM (1963).
Buffered glutaraldehyde — a new chemical
sterilizing solution. Am J Hosp Pharm
20:459-65.
Jordan \VP Jr, Dahl MV, Albert HL (1972).
Contact Dermatitis from Glutaraldehyde.
Arch Dermatol 105: 94-95.
Jordan WP Jr, Dahl MV, Albert HL (1972).
Contact Dermatitis from Glutaraldehyde.
Arch Dermatol 105: 94-95.
4-
Nethercott JR et al (1988). Occupational
contact dermatitis due to glutaraldehyde in
health care workers. 18:193-6.
5.
Werley MS, Burleigh-Flayer HD, Ballantyne
B (1995). Respiratory Peripheral Sensory'
Irritation and Hypersensitivity Studies with
Glutaraldehyde Vapor. Toxicology and
Industrial Health 11 (5): 489-501.
6.
7.
Di Stefano F et. al (1998) Occupational
asthma due to glutaraldehyde, Monaldi
Archives of Chest Diseases 53:50-5.
Corrado OJ, Osman J, Davies RJ (1986).
Asthma and Rhinitis after exposure to
Glutaraldehyde in Endoscopy Units. Human
Toxicology’ 5 (5): 328-8.
8.
Chan-Yeung M, McMurren T, CatonioBegley F, Lam S (1993). Occupational asth
ma in a technologist exposed to glutaralde
hyde. J Allergy Clin Immunol 91(5). 974-8.
9.
Gannon PFG et al (1994) Occupational
asthma due to glutaraldehyde and formalde
hyde m endoscopy and x ray departments.
Thorax 50: 156-159,
10. Di Stefano F, Siriruttanapruk S, McCoach J,
Sherwood Burge P (1999) Glutaraldehyde:
an occupational hazard in the hospital set
ting. Allergy 54:1105-1109.
11. ACGIH (1998). Glutaraldehyde. Draft
chemical summary and recommendations.
American Congress of Government
Industrial Hygienists. November 16, 1998
12. MMWR. Epidemiologic notes and reports:
Symptoms of irritation associated with expo
sure to glutaraldehyde. Colorado. April 3,
1987/36(12): 190-1.
13. Ziem G, McTamney J (1997). Profile of
Patients with Chemical Injury and
Sensitivity. Environmental Health
Perspectives 105 (supplement 2): 417-36.
14. Rutala WA (1996) APIC Guideline for
Selection and Use of Disinfectants.
American Journal of Infection Control
24:313-42.
15. CDRH (2000). Sterilants and High Level
Disinfectants Cleared by FDA in a 510(k) as
of January 28, 2000 with General Claims for
Processing Reusable Medical and Dental
Devices. Center for Devices and
Radiological Health Office of Device
Evaluation, Division of Dental, Infection
Control and General Hospital Devices.
Internet Download on 7/11/00.
www. fd a .gov/c d r h/od e/ge rm 1 a b. h t m I
16. Royal College of Nursing (2000). Is There
an Alternative to Glutaraldehyde? A Review
of Agents used in Cold Sterilisation. Royal
College of Nursing, Working Well Initiative.
November, 2000.
17. Crow S (1993). Peracetic Acid - Asking the
Right Questions. Today’s O.R. Nurse,
May/June 1993: 47 - 49.
18. ACGIH (1999). Documentation of the
Threshold Limit Values and Biological
Exposure Indices, 6th Ed. American
Conference of Governmental Industrial
Hygienists; Publication 0206, Cincinnati,
OH.
19. Rutala WA (1996) APIC Guideline for
Selection and Use of Disinfectants.
American Journal of Infection Control
24:313-42.
20. Dartmouth Hitchcock Medical Center
(DHMC). Glutaraldehyde Waste
Minimization Report. Unpublished report,
1997. Lebanon, New Hampshire.
Without Harm
1755 S Street, NW
Unit 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org .
Tlie PCF certification mark and term arc the sole property of the Chlorine Free
Products Association and are only used by authorized and certified users.
Needlestick
injuries
Fact Sheet
Prepared cooperatively by the
American Nurses Association and
the Intravenous Nurses Society
March 2001
<13
c
The Problem
The Solution = Prevention
An estimated 600,000 - 800,000
needlestick injuries (nsi) occur annual'
ly in the United States.1 About half of
these injuries go unreported. An aver'
age hospital incurs approximately 30
worker nsi per 100 beds per year
according to the Exposure Prevention
Information Network (EPINet) exposure surveillance data from the
International Health Care Worker
Safety Center at the University of
Virginia-Charlottesville.2 Most reported
nsi involve nursing staff, but lab staff,
physicians, housekeepers, and other
health care workers are also injured.’
Some of these injuries expose workers to
bloodborne pathogens, including hepati
tis B, hepatitis C and HIV Infection
with any of these pathogens is potential
ly life-threatening.
The only real solution is prevention.
Preventing exposure to blood by pre
venting needlestick injuries will pre
vent disease.
The risk of infection from hepatitis is
much greater than the risk of HIV, and
while there is an immunization to pre
vent transmission of hepatitis B, and
post-exposure prophylaxis and expen
sive treatment for HIV, there is cur
rently no recommended prophylaxis or
effective treatment for hepatitis C.
Seventy-five percent of individuals
infected with hepatitis C will become
chronically infected. The eventual
outcome of hepatitis C is liver failure.
The only treatment for liver failure is a
liver transplant.
According to the Centers for Disease
Control and Prevention (CDC), up to
86% of needlestick injuries can be pre
vented by using safer needlestick
devices. ’ A combination of work prac
tice controls, safety education, sharps
disposal containers and safety devices
can reduce the risk of bloodborne
exposures by 94%.5
Safer needle devices are cost-effective.
A safety needle costs about 28 cents
more per needle, but the extra expense
is minimal compared to the approxi
mately $1 million facilities will spendfor a needlestick inury that results in a
serious infection.6
In 1992, the US Food and Drug
Administration (FDA) recommended
that health care institutions eliminate
the use of sharps for supplemental (pig
gyback) administration of fluids into
existing intravenous (IV) lines7 as an
unnecessary hazard. Despite this rec
ommendation, only 2/3 of the hospitals
in the US implemented IV needleless
systems, and by 1999, only 15% of hos
pitals had implemented safer needle
devices for injection, phlebotomy and
intravenous access.0
In 1999, the FDA, the National
Institute for Occupational Safety and
Health (NIOSH) and the
Occupational Safety and Health
Administration (OSHA) recommend
ed the elimination of glass capillary
tubes used for blood collection and
replacement with plastic capillary' tubes
to prevent injury7 and exposure to
blood.9
In 2000, following the passage of simi
lar laws in 17 states, Congress passed
the Needlestick Safety and Prevention
Act which amended the 1991 OSHA
Bloodborne Pathogens Standard (BPS)
to require the use of sharps with engi
neered sharps injury protections also
known as safer needle devices.10 This
law requires the involvement of front
line health care workers in the evalua
tion, selection and implementation of
safety devices. This is essential for
clinically appropriate purchasing deci
sions and eases the process of imple
mentation. Training is necessary to
involve workers in the exposure con
trol program and is required annually
by the OSHA BPS. In addition, the
law requires device-specific data col
lection regarding the cause of the
needlestick injury (29 CFR 1910).
Desirable Characteristics
of Safety Devices"
E
3
»
3
All institutions should utilize the safer
medical devices that are appropriate,
commercially available, and effective.
The following categories of sharps
have commercially available safety'
devices.12
Blood collection
(phlebotomy) devices
3
Shielded, self-blunting, or retract
ing needles for vacuum tub phle
botomy sets
n
Plastic vacuum/specimen tubes
resistant to breakage
s
Shielded, self-blunting, or retract
ing winged-steel needles
®
Blood gas syringes with a hinged
needle recapping device
Retracting finger/heelstick lancets
3
Unbreakable plastic capillary tubes
3
Hemoglobin readers that do not
use capillary' tubes or require cen
trifuge of the sample
3
Adapters for needleless IV systems
The device preferably works pas
sively (i.e., it requires no activation
by the user). If user activation is
necessary, the safety feature can be
engaged with a single-handed
technique and allows the workers’
hands to remain behind the
exposed sharp.
3
The user can easily tell whether
the safety feature is activated.
Injection devices
The safety feature cannot be deac
tivated and remains protective
through disposal.
3
The device performs reliably.
3
The device is easy to use and prac
tical.
3
Safety Sharps
Available on the Market
The device is needleless.
The safety feature is an integral
part of the device.
The device is safe and effective for
patient care.
Suture needles and
scalpel blades
Criteria for device selection is avail
able from the Training for rhe
Development of Innovative Control
Technologies (TDICT) Project at
www.tdict.org/cnteria.html
IV Catheter insertion devices
Shielded or retracting stylets
■
Shielded and retracting needles
3
Recapping with sliding
sheath/sleeve (OSHA BPS pro
hibits recapping)
Blunt needle/cannula to access the
injection port
3
Valve or stopcock to be used with a
syringe (without a needle)
°
Protected or recessed needles
3
Pre-filled medication cartridge with
safety' needles
Rounded tip scalpel blades
3
Retracting scalpel blades
«
Shielded scalpel blades
3
Disposable scalpels (blade removal
not necessary)
■
Quick release blade handles
Two web databases provide informa
tion regarding the safety devices cur
rently on the market. The University
of Virginia -Charlottesville
International Center for Healthcare
Worker Safety' maintains a list of prod
ucts commercially available in the
above categories on the Web at
www.med.virginia.edu/~epinet.
The California Department of Health
Services Sharps Injury Control
Program maintains a web site at
www.dhs.ca.gov/sharps
Checklist for
Compliance with
Amended Bloodborne
Pathogens Standard
(as amended by the
Needlestick Safety and
Prevention Act of 2000)
The following questions will assist in
determining compliance with the law
(adapted from the ANA Checklist for
Compliance at www.needlestick.org)
3
Does a written Exposure Control
Program (ECP) exist?
3
Has a hard copy of the ECP been
made available to employees or
their representatives within 15
working days of a request for one?
3
Is the ECP reviewed and updated
annually or more frequently when
ever new or modified procedures
are adopted or whenever employ
ee positions are revised in such a
way that creates new potential
exposures?
3
Does the annual review of the ECP
include a review of the most recent
technological advances?
IV delivery systems
3
■
a
■
a
s
Does the review of safety devices
include the involvement of front
line health care workers (non
managerial employees responsible
for direct patient care), which is
required in device evaluation and
selection, with evidence of this
participation documented in the
ECP?
Are safer needles and other sharps
with integrated safety features being
used when medically appropriate?
Are purchasing decisions based on
the safest and most effective option
as opposed to the least expensive?
Have frontline health care workers
received interactive training on the
P use of safer devices from a knowl
edgeable person; been informed of
the location of the ECP and the
procedures to follow if an exposure
occurs?
D
4.
Are needleless or shielded needle
IV line access products provided?
a
u
3.
Does post-exposiire follow-up that
conforms to the CDC guidelines
for testing and prophylaxis occur
within two hours of the exposure?
Is there a sharps injury log updated
regularly with the details of all
needlestick injuries, including
device brand and type?
Quality care can be assured only when
health care workers are safe from the
risk of disease and death caused by
innecessary needlesticks. Front-line
lealthcare workers should not have to
risk their lives while saving the lives of
their patients.
Notes
1.
US Department of Health and Human '
Services. National Institute for
Occupational Safety and Health (NIOSH).
NIOSH ALERT Preventing Needlestick
Injuries in Health Care Settings.
Publication No. 2000-108. 2000.
2.
EPINet. Exposure prevention information
network data reports. University of
Virginia: International Health Care Worker
Safety Center, 1999.
5.
US Department of Health and Human
Services. National Institute for
Occupational Safety and Health (NIOSH).
NIOSH ALERT Preventing Nccdlcstick
Injuries in Health Care Settings.
Publication No. 2000-108. 2000.
Centers for Disease Control and Prevention
Evaluation of safety devices for preventing
percutaneous injuries among health care
workers during phlebotomy procedures —
Minneapolis-St. Paul, New York City, and
San Francisco, 1993-1995. MMWR
46(2):21-25.
Jagger J. Reducing occupational exposure to
bloodborne pathogens: where do we stand a
decade later? Infect Control Hosp
Epidemiol 17 (9) .-5733-575,1996.
6.
Pugliese G and Salahuddin M. cds. Sharps
Injury Prevention Program A Step-By-Step
Guide. Chicago, Illinois: American
Hospital Association, 1999.
7.
US Department of Health and Human
Services. Food and Drug Administration
(FDA). FDA Safety Alert. Needlestick and
Other Risks from Hypodermic Needles on
Secondary I.V Administration SetsPiggyback and Intermittent I V, April 16,
1992. Available: www.oshaslc.gov/SLTC/needlestick/fdalctter.html
8.
Pugliese G and Salahuddin M. cds. Sharps
Injury Prevention Program A Step-By-Step
Guide. Chicago, Illinois: American
Flospital Association, 1999.
9.
US Department of Health anti Fluman
Services. FDA. Glass Capillary' Tubes:
10. Federal Register, vol. 66, January 18, 2001,
pp5318-5325
11. US Department of Health and Human
Services. National Institute for
Occupational Safety and Health (NIOSH).
NIOSH ALERT Preventing Needlestick
Injuries in Health Care Settings.
Publication No. 2000-108. 2000.
12. McCormick R. “Selecting Safety Products
for Evaluation” m Pugliese G and
Salahuddin M eds. Sharps InjuryPrevention Program A Step By Step Guide.
Chicago, Illinois; American Hospital
Association, 1999.
Resources
OSHA. OSHA Directives 2-2.44D.
Enforcement Procedures for the occupational
exposure to bloodborne pathogens. Washington,
DC: U S. Department of Labor, Occupational
Safety and Health Administration. 1999
(www.osha-slc.gov/OshDoc/Directivc_data/
CPL_2-2_44D.html).
Wilburn S. Know Your Rights: Ensuring your
employer’s compliance with federal nccdlcstick
law. American Journal of Nursing. Vol 101,3:
90. March 2001.
Cteanwg
a
pftate
Fact Sheet
Chemical use in hospitals contributes
to poor air quality and has been impli
cated in the increase of worker respira
tory ailments such as asthma and
Reactive Airway Dysfunction Syndrome
(RADS). Exposure to and contact with
cleaning chemicals can also cause eye,
nose and throat irritation, skin rashes,
headaches, dizziness, nausea and sensiti
zation. According to the Massachusetts
Department of Public Health (DPH),
the most commonly reported occupa
tional asthma-causing agent is poor
indoor air quality’.
Good air quality results in an environ
ment where workers feel healthy and
comfortable and as a result, are more
productive. This decreases both costs
and liabilities. Adequate ventilation in
relation to environmental cleaning
products and processes is a major fac
tor in good air quality.1 By carefully
choosing environmentally sound clean
ing chemicals, cleaning methods and
cleaning equipment, U.S. businesses
could realize a productivity gain of $30
to $150 billion annually and a 0.5% to
5% increase in worker performance.
According to the American Lung
Association (ALA), asthma is the most
prevalent occupational lung disease in
developed countries.2 Cleaning and
disinfecting chemicals such as ammo
nia, chlorine, cleaning detergents, eth
ylene oxide, pesticides, and sodium
hydroxide, are listed by the DPH as
causing RADS.’ Nursing, teaching and
office work are the occupations most
likely to report problems with indoor
air quality. DPH statistics from 19931998 note that nurses have the highest
number of reported cases of work-relat
ed asthma, and indicate that health
care is the industry with the most cases
of work-related asthma. The most fre
quently reported exposures in health
care were to latex, poor indoor air
quality', and toxic cleaning products.4
Toxic cleaning chemicals contribute to
poor indoor air quality and worker ill
nesses through a combination of the
product selected and the processes uti
lized to apply the chemicals.
Product
Disinfectant chemicals
Disinfectants used in hospitals such as
quaternary’ ammonium compounds,
phenols, and bleach are registered with
the EPA as pesticides. These toxic
chemicals are used for routine cleaning
on every surface in the hospital envi
ronment. Health effects from long-term
exposure to quaternary’ ammonium
compounds include occupational asth
ma and hypersensitivity syndrome.5-6
Floor stripping and
polishing chemicals
Floor strippers contain chemicals that
can seriously harm the user and may
also affect the building occupants.
Chemicals in these products include
diethylene glycol ethyl ether, aliphatic
petroleum distillates and nonyl-phenol
ethoxylate, ethanolamine (a known
sensitizer), butoxyethanol, and sodium
hydroxide (lye).
Health care workers and others
exposed to floor stripping and floor pol
ishing chemicals experience headaches,
eye irritation, dizziness, nausea, difficul
ty concentrating, fatigue, wheezing,
coughing, asthma attacks, respiratory’
infections, hypersensitivity' pneumoni
tis, and nose, throat and skin irritation.
If exposure continues, irreversible lung
damage and the formation of fibrous
tissue (fibrosis) may occur making
breathing more difficult.
Scented cleaning chemicals
The use of unscented cleaning chemi
cals is recommended to improve indoor
air quality. The Archives of
Environmental Health note that some
humans exposed to fragrance products
might experience some combination of
eye, nose and/or throat irritation; respira
tory difficulty'; possibly broncho-constric
tion, or asthma-like reactions; and cen
tral nervous system reactions (eg. dizzi
ness, incoordination, confusion, fatigue).7
Process
Inadequate ventilation
Inadequate ventilation, reducing the fre
quency and volume of air exchanges, or
climate controls designed to save energy,
increases the concentration of chemicals
in indoor air. Extensive and complex
cleaning projects (floor stripping, bur
nishing, rug cleaning) are often carried
out on the overnight shift in hospitals,
when fewer people are around, but also
when ventilation is reduced to save ener
gy. Additions, newer hospitals or remod
eled areas are often very tight buildings
with little or no natural ventilation and
may have windows that do not open to
allow fresh air intake to dilute these
chemicals. Ventilation and fresh air
exchanges should be increased when
these projects are carried out.
Mixing of Chemicals
Cleaning chemicals are often purchased
in concentrated solutions that require
mixing and/or dilution by the employee
who is responsible for application. It has
been noted that when adverse health
effects are suffered by workers, the con
centration (or mixtures) of these prod
ucts is often incorrect. This may indicate
a problem with training, language skills
or worker supervision.
When certain cleaning chemicals are
mixed together synergistic effects may
occur. This means that the interaction of
two or more of these chemicals produces
a health effect greater than that of the
individual chemical alone. For example,
if a quaternary ammonium compound is
use in combination with a bleach cleaner,
a toxic gas called chloramine forms and
is released into the air.
Application methods
of cleaning chemicals
The use of spray bottles, aerosol cans,
and mechanized equipment, such as floor
burnishers, buffers, and carpet washers,
increase the airborne concentration of
cleaning chemicals as particulate matter
becomes aerosolized and suspends in the
breathing zone of operators and building
occupants. Spray bottles should be
replaced with a pour and wipe applica
tion process. Floor burnishers and
buffers should have an enclosed system
with a filter (scrubber) to capture chemi
cal vapors and particulate matter that is
generated during the burnishing process.
These changes will contribute to the
reduction of the aerosol concentration of
these cleaning chemicals and their by
products. These changes decrease air
contamination and contribute to
improved indoor air quality and the
health and comfort of all the building
inhabitants.
Resources
The following articles and guidelines will
assist you in modifying the use and selec
tion of cleaning chemicals for improved
indoor air quality' and a safer healthier
work environment.
° A detailed report from INFORM, Inc.
can be obtained by contacting Lara
Sutherland via email at
sutherland@informinc.org. This
report is an in-depth look at the
problems with cleaning chemicals and
possible solutions.
a
A list of environmentally preferable
products, also noted as the best in
class, The OSD Update, 99-31, can
be obtained from the Massachusetts
Operational Services Division, at One
Ashburton Place, Room 1017, Boston,
MA 02108. These products have
been evaluated and accepted using a
variety of environmental and health
concerns as criteria.
a
The Janitorial Pollution Prevention
Project provides quick reference and
worksheets on a variety of cleaning
processes and materials focusing on
safe and healthy work practices.
www. wes tp2 ne t .org/ J an i to ri al/j p4. h tm
References
1.
Fisk, William and Arthur Rosenfield.
“Improved Productivity and Health from
Better Indoor Environments,” Center for
Building Science Newsletter (Now
Environmental Energy Technologies
Newsletter). Lawrence Berkley Labs. Summer
1997. p.5. Available at http://eetd.lbl.gov/
Bookstore.htm
2.
American Lung Association, “Occupational
Hazards,” 2000, p.3.
www.Iungusa.org/air/airOO_occupation.hcml
3.
MA DPH SENSOR Occupational Lung
Disease Bulletin. November, 2000.
4.
MA DPH SENSOR Occupational Lung
Disease Bulletin. January, 2000.
5.
Bernstein, J A Combined Respiratory and
Cutaneous Hypersensitivity Syndrome to Quat
Amines. Jnl Allergy Clin Immunol 1994;
Vol.94, No.2, pp 257-259.
6.
Personal Communication; Amy Smoker, MS.
Benzlkonium Chloride Fact Sheet. National
Antimicrobial information Network, Oregon
State University, http://nain.orst.edu
800 447-6349,
1-
7.
Drs. Rosalind C. Anderson and Julius H
Anderson, “Acute Toxic Effects of Fragranced
Products". Archives of Environmental Health
53(2): 138-146 (1998).
8.
California Office of Environmental Health
Hazard Assessment Fact Sheet. Health Effects
of Diesel Exhaust. August, 2000.
www.oehha.ca.gov/air/diesel_exhaust/
factsheet.html
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.nohann.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care, for additional copies of this or other pub
lications included in the kit. or to find out how to get a complete kit.
visit Health Care Without Harm on the Web at www.noharm.org.
l^jSOYINK
The PCF ccrimcation mark and term are lhe s'!c property <4 the ChhniK Free
Product) /Vsociation and arc only
by auiluTired .mJ certihcd uxts.
Going Green:
A Resource
Kit for
Pollution
Prevention in
Health Care
Table of
Contents
Overview - BLUE
1
1Introduction
213
1-
Who We Are
Memorandum of Understanding Between the US EPA and the American Hospital
Association
415
1-
Hospitals for a Healthy Environment (H2E) (Under development)
Sustainable Hospitals Project (Under development)
6
1-
List of Resolutions/Ordinances on PVC, Dioxin & Mercury
Mercury - RED
1
2The Mercury Problem --Fast Facts
223
2-
Making Medicine Mercury Free
List of Mercury-Containing Items in a Hospital Setting
24
5
2-
Thermometer Fact Sheet
How to Hold a Mercury Thermometer Rnundup
627
22-8
Battery Roundups: Get Charged!
List of Mercury Recycling Companies
"Mad As A Hatter" Campaign for a Mercury-Free NIH (Under development)
2-9
Replacing Mercury Sphygmomanometers (Under development)
Dioxin, PVC & DEHP - PURPLE
1
3-
Dioxin, PVC & Health Care
233
3-
What's Wrong With Incineration?
Alternative Technologies Report Order Form
4
3-
Reducing PVC Use in Hospitals
536
3-
PVC Alternatives
DEHP Exposures During the Medical Care of Infants: A Cause for Concern
7
3-
A Summary of the Expert Panel Report of the National Toxicology Program on DEHP
and its Risks to Human Reproduction
3-8
A Summary of the FDA Safety Assessment of DEHP released from PVC Medical
Devices (Under development)
3-9
PVC/DEHP Resource List
Waste Minimization - ORANGE
1
4Waste Minimization, Segregation and Recycling in Hospitals
I
o
42
3
4-
10 Steps to Consider Red Bag Reduction Program
Guidelines for Optimizing Waste Segregation
4
4-
Disposables and their Alternatives
45
6
47
4-
Reach for Unbleached Paper
Recycling Fact Sheet
Waste Minimization Resources
Environmentally Preferable Purchasing - GREEN
1
5Environmentally Preferable Purchasing How-to Guide
4, X)
e
253
5-
Sample Letter to Group Purchasing Organizations
Model Contract Language for the Elimination/Phase-out of PVC Medical Products
(Under development)
Worker Health & Safety - YELLOW
1
6Latex Allergy in Health Care Fact Sheet
2
6Needlestick Fact Sheet
3
610 Reasons to Eliminate Gluturaldehyde
6-4 Cleaning Chemical Use in Hospitals Fact Sheet
An unfortunate irony of the current
health care system is that certain
practices pose threats to public
health and the environment.
Without Harm
Pub 1-01 This publication is part of Going Green: A Resource Kit for
Pollution Prevention in Health Care Visit Health Care Without Harm on
the Web at www.nohiirm.org for more information.
October 15, 2001
Fortunately, many of these risks can be
eliminated through fairly simple
changes in the way a hospital operates
and in the materials it purchases. For
example, public and occupational
health risks from mercury use can be
eliminated by making sure that mercu^containing medical devices and
products are phased out, and that
existing ones, when retired, are not
incinerated. By replacing existing mer
cury' devices with non-mercury alterna
tive devices for the future, this avoid
able risk is eliminated.
unions, environmental groups and
community' organizations - has put
together this resource kit to assist
health care providers/administrators in
their efforts to reduce health care
industry' pollution from their facilities.
A “work in progress,” this Resource Kit
contains steps that range from the sim
ple to the complex, but all will have a
measurable impact on your facility’s
environmental performance.
The information available to help
health care facilities improve their
environmental performance is con
stantly evolving. HCWH works to
update its resources in a timely fashion.
Look for the latest version of these
resources as well as new materials on
our web site at www.noharm.org.
Awareness of health care industry pol
lution and the need to engage in
improved material purchasing and
waste management policies has led to
the creation of a number of pollution
prevention partnerships. In 1998 the
American Flospital Association and
the U.S. Environmental Protection
Agency signed a Memorandum of
Understanding to prevent the release
of persistent, bio-accumulative toxic
chemicals released by the health care
industry'. The MOU established a
series of goals and timetables to elimi
nate mercury releases, reduce waste,
and minimize the production of persist
ent, bio-accumulative toxins.
This Resource Kit - and the steps con
tained in it - draw upon the collective
wisdom and experience of health care
professionals and others who have
been successful at a number of facilities
around the country'. Many of the sug
gestions have the additional benefit of
being economically sound as well,
which is particularly welcome in an era
of cost containment.
Many hospitals and health care systems
across the U.S. are already making
great strides toward reducing the envi
ronmental impact of the health care
industry', but more work needs to be
done. Health Care Without Flarm
(FICWH) - an international collabora
tion of hospitals and health care sys
tems, medical professionals, labor
HCWH strongly encourages you to use
the information here to help reduce
the health care industry's environmen
tal impact. Together we can make a
profound difference in reducing envi
ronmental harm from health care prac
tices, and demonstrate that our profes
sions, industry, and our partners can
work together for a healthier future.
Who We Are
The Health Care Without Harm coali
tion is a broad-based international
campaign to address the environmental
impacts of health care, without com
promising worker safety or patient care.
Our efforts include:
D
Without Harm
The Campaign for
Environmentally Responsible
Health Care
advocating for policies to eliminate
the indiscriminate incineration of
medical waste,
n
changing purchasing and materials
management practices of hospitals
and purchasing groups,
n
promoting policies and procedures
that work toward the minimization
of waste volume and toxicity,
n
supporting local campaigns to
oppose medical waste incinerators,
D
researching and advocating safer
waste disposal alternatives, and
Q
educating the broader public about
dioxin, mercury, and endocrine-dis
rupting chemicals and the health
care industry’s contribution to
these problems.
Since its inception in 1996, the mem
bership of the Health Care Without
Harm (HCWH) campaign has grown
from an initial 28 founding organiza
tions, to more than 340 organizations
in 37 countries. The coalition has
attracted the attention of major health
care systems, regulatory bodies and
health manufacturers of medical prod
ucts. Membership includes more than
100 health care facilities, and numer
ous organizations of health profession
als: the American Nurses Association;
the American Public Health
Association; the Ambulatory Pediatrics
Association; the Oncology Nursing
Society; and many more.
The Campaign is a coalition of tradi
tional health care organizations, as well
as religious constituencies, labor
unions, health-affected constituencies
(cancer groups, endometriosis groups,
children’s health groups, etc.) and
environmental groups, who agree that
as providers and consumers of health
care, we all have a place in a campaign
that makes explicit links between envi
ronmental contamination and public
health. Health Care Without Harm is
an attempt to bring these important
constituencies together in a broad
effort on an issue that affects the
health of every man, woman and child.
The Campaign understands that health
professionals are not intentionally
employing products or practices; they
are simply not aware of the links
between these products/practices in
terms of environmental contamination
and illnesses in the general population.
In fact, as physicians, nurses and other
health professionals have learned about
the health risks of mercury, dioxin and
other pollutants emitted by the use
and/or disposal of products used in
health care, they have generally sup
ported actions that will reduce or elim
inate that pollution while providing
safe, effective patient care.
Groups that join the campaign do not
contribute dues. Membership in
Health Care Without Harm is based
upon an organizational commitment to
the mission and goals of the campaign,
and a desire to participate fully in help
ing to achieve them. Health Care
Without Harm is funded entirely by
foundations and individuals. The cam
paign does not accept financial support
from manufacturers of medical
supplies and does not endorse specific
products.
Memorandum
of Understanding
between the
American
Hospital
Association
& the UoSo
Environmental
Protection
Agency
On June 24, 1998, a landmark agree
ment was put together by the
American Hospital Association (AHA)
and the United States Environmental
Protection Agency (EPA). The
Memorandum of Understanding
(MOU) set new goals for hospital pol
lution prevention over the next five
years, and brought together a stake
holders’ council to enforce the provi
sions of the MOU. Health Care
Without Harm (HCWH) was an active
participant in the preparation of the
agreement, and sat on the AHA
Leadership Council.
The MOU set 10 action steps for the
council to focus on over a five-year
period. Two of the top priorities are the
virtual elimination of mercury-containing waste from the hospital waste
stream by the year 2005, and the goal
of achieving a thirty-three percent
(33%) reduction in total waste volume
in all hospitals by 2005 and an overall
goal of achieving a fifty percent (50%)
reduction by 2010.
The ten points of the plan are as follows:
1. Virtual Elimination of Mercury
Waste.
2. Total Waste Volume Reduction.
3. Seminars.
4. Software Distribution.
5. Industry' P2 Information.
6. Review of Industry' P2 Information.
7. Chemical Waste Minimization.
8. Ethylene Oxide and PBT Pollutant
Information.
9. Industry Input on U.S. EPA
Guidance.
10. AHA Environmental Leadership
Council.
11. Awards/Recognition.
Hospitals for a Healthy Environment
(H2E) was adopted as the title for this
effort. In September 2001, H2E
became a partnership of the AHA,
EPA, HCWH and the American
Nurses Association (ANA). An H2E
listserve has been developed. Join the
H2E listserv to share and learn techni
cal information, find educational tools
and identify practical strategies for
mercury' elimination and discuss other
pollution prevention and waste mini
mization issues. For information on
how to become an active participant in
the H2E process, see their website at
www.h2e-online.org.
The Memorandum
1.0 INTRODUCTION.
This Memorandum of Understanding
(“MOU”) is made between the United
States Environmental Protection
Agency (“U.S. EPA”) Office of
Prevention, Pesticides and Toxic sub
stances (“OPPT”), U.S. EPA Region 5
and the American Hospital
Association (“AHA”). Throughout this
MOU, any reference to “U.S. EPA”
shall include both OPPT and Region 5
and any reference to “AHA” shall refer
to AHA and its Personal Membership
Groups (“PMGs”). U.S. EPA and AHA
are referred to herein as “the Parties”
to this MOU.
1.1 The Parties intend by this MOU to
establish a mutually beneficial
public/private partnership.
1.2 This MOU will address the basic
relationship, roles and responsibilities
of the Parties but leaves for later agree
ment the more precise terms that will
constitute the substance of the part
nership.
2.0 PURPOSE.
The ALIA consists primarily of health
care provider organizations across the
United States. The Parties enter into
this MOU for the primary' purpose of
transferring to AHA institutional
members, PMG personal members and
other health care professionals techni
cal information on Pollution
Prevention (“P2”) opportunities that
exist with respect to waste generated
by the health care industry. The
Parties’ believe that this information
transfer will provide the health care
industry with enhanced tools for mini'
mixing the production or persistent, bio
accumulative and toxic (“PBT”) pollu
tants and reducing the volumes of
waste generated. Such reductions are
beneficial to the environment and will
reduce the waste disposal costs incurred
by the health care industry'. The Parties
to this MOU hereby affirm the
Congressional goals and principles set
forth in the Pollution Prevention Act
(“PPA”), 42 U.S.C. 13101 through
13109, particularly the goal of reducing
the generation of pollution at its source,
preferentially to the recycling, treat
ment and/or disposal of such waste.
3.0 AUTHORITY.
Section 6604(b)(5) of the PPA, 42
U.S.C. 13103(b)(5), directs U.S. EPA,
among other things, to facilitate the
adoption of source reduction tech'
niques by businesses, including the dis
tribution of source reduction informa
tion to businesses.
4.0 ROLES AND RESPONSIBILI
TIES OF THE PARTIES.
The Parties intend to undertake the
following activities pursuant to this
MOU:
1. Virtual Elimination of Mercury’
Waste. The Parties intend to work
together to develop a Mercury' Waste
Virtual Elimination Plan that will set
forth a strategy' for achieving the goal
of virtually eliminating mercury-containing waste from the health care
industry' waste stream by the year
2005.
2. Total Waste Volume Reduction. The
Parties intend to work together to
develop a Model Waste Volume
Reduction Plan that will assist in
reducing the total volume of all wastes
(including both regulated and non-regulated waste) generated by the health
care industry’, with an initial goal of
achieving a thirty-three percent (33%)
reduction in all hospitals by 2005 and
an overall goal of achieving a fifty' per
cent (50%) reduction by 2010.
3. Seminars. The Parties intend to co
sponsor a series of Health Care
Industry Waste Management Seminars
(“Seminars”) to be held at various
locations across the United States.
The Seminars will be the primary' vehi
cle by which technical information on
P2 opportunities will be transferred to
the health care professionals, and will
focus upon transferring technical infor
mation related to decreasing health
care industry' waste volume, minimiz
ing the production of PBT pollutants,
improving waste stream segregation,
reducing waste management costs and
ensuring regulatory' compliance for
regulated waste streams.
4. Software Distribution. In order to
facilitate the successful completion of
the Seminars and the virtual elimina
tion of mercury-containing waste, U.S.
EPA intends to provide for distribution
at the various Seminars up to 300
copies of the software program entitled
“Mercury' In Medical Facilities” that
has been developed by Purdue
University with assistance from the
Region 5 Software Development Unit
(“SDU”). Purdue University' maintains
a copyright on this software program,
but, insofar as the software was devel
oped with Federal Government assis
tance, the software may be freely
copied and disseminated. The Parties
will mutually decide how the up to 300
total software copies will be distributed
among the various Seminars.
5. Industry' P2 Information. AHA
intends to develop baseline informa
tion on the P2 activities of the health
care industry and to monitor P2
progress over time. To obtain this
information, AHA will develop, with
review and comment by U.S. EPA, an
information questionnaire to be dis
tributed to health care professionals by
AHA at various times in the future.
The first distribution will be used to
determine the baseline P2 information
and subsequent distributions will be
used to monitor industry P2 progress.
AHA will gather all responses to the
questionnaires. Insofar as U.S. EPA
will not be sponsoring the distribution
of the questionnaire, the distribution
of the questionnaire is not subject to
the requirements of the Paperwork
Reduction Act (“PRA”), 44 U.S.C.
3501 through 3520.
6. Review of Industry P2 Information.
Throughout the duration of this
MOU, the Parties intend to work
together to review and compile the
information obtained from the baseline
and progress questionnaires (Item #5).
U.S. EPA agrees that, unless required
by law, the identity of any survey par
ticipant need not be revealed by AHA
to U.S. EPA. From this information,
the Parties will be able to disseminate
more effectively P2 information and to
monitor the success of the Mercury'
Waste Virtual Elimination Plan (Item
#1) and the Model Waste Volume
Reduction Plan (Item #2).
(
7. Chemical Waste Minimization. The
Parties intend to work together to
develop, for various kinds of chemical
waste, a Model Chemical Waste
Minimization Plan (“Model Plan”).
The first Model Plan will pertain to
mercury-containing waste (“Model
Plan For Mercury'”). The Model Plan
For Mercury is presently being devel
oped by the State of Illinois with assis
tance from U.S. EPA. When that plan
is completed, U.S. EPA, with com
ments from AHA, will make such
modifications to the Model Plan For
Mercury' as are necessary' to reflect
current knowledge, best management
practices and any other circumstances
experienced by the health care indus- (
try. Other chemical wastes will be
addressed by future Model Plans.
AHA intends to disseminate each
Model Plan to as wide an audience in
the health care industry as is reason
ably possible. Both AHA and U.S.
EPA intend to make each Model Plan
available to the public on their respec
tive Internet home pages. Each such
Internet presentation shall properly
reflect the relative contributions of the
Parties and any third party (such as
the State of Illinois with respect to the
Model Plan For Mercury) to the devel
opment of the particular Model Plan.
8. Ethylene Oxide and PBT Pollutant
Information. The Parties intend to
3. By mutual agreement, which may be
either formal or informal, the Parties
may modify the list of intended activi
ties set forth in Paragraph 4-0 above
and/or determine the practical manner
by which the goals, purposes and
activities of this MOU will be accom
plished. However, any modification to
7. AHA understands and acknowl
edges that, as an institution of the
Federal Government, U.S. EPA has a
duty to refrain from providing any
commercial entity an exclusive privi
lege without receiving payment there
fore and, as a consequence, that U.S.
EPA’s relationship with AHA in no
way affects, alters or otherwise con
strains U.S. EPA’s right to provide sim
ilar (or identical) services to, or estab
lish similar (or identical) relationships
with, any other entity.
8. AHA understands that U.S. EPA’s
participation in this MOU does not
constitute an endorsement, express or
implied of (a) any policy advocated by
AHA, the Council or any stakeholder;
or (b) any good or service offered or
sold by AHA, the Council or any
stakeholder.
9. Insofar as U.S. EPA’s participation in
this MOU consists of rendering tech-
OF
2. Either Party may unilaterally with
draw at any time from this MOU by
transmitting a signed writing to that
effect to the other Party. This MOU
and the public/private partnership cre
ated thereby shall be considered termi
nated sixty (60) days from the date the
non-withdrawing Party' actually
receives the notice of withdrawal from
the withdrawing Party7.
6. AHA agrees that it does not expect,
nor will it ever seek to compel from
U.S. EPA in any judicial forum, the
payment of money, services or other
thing of value from U.S. EPA based
upon the terms of this MOU. The
foregoing provision does not in any
way affect any legal rights accruing to
AHA by virtue of any other law, con
tract and/or assistance agreement.
MEMORANDUM
4.1 The Parties understand that other
organizations and/or coalitions who
promote environmentally responsible
practices have a vested interest in the
goals described in this MOU.
Furthermore, the Parties recognize that
1. Each Party pledges in good faith to
go forward with this MOU and to fur
ther the goals and purposes of this
MOU, subject to the terms and condi
tions of this MOU. The Parties shall
attempt to resolve disputes through
good faith discussions.
5. Nothing in this MOU shall be con
strued to authorize or permit any vio
lation of any Federal, State or local
law, including, but not limited to, any
environmental law administered
and/or enforced by U.S. EPA, by any
person, including, but not limited to,
any health care provider organization.
U N D E R S T A N D IN G B E T W E E N T H E A M E R IC A N H O S P IT A
E N V IR O N M E N T A L P R O T E C T IO N A G E N C Y
11. Awards/Recognition. The Parties
intend to work together to determine
national “success stories” of the imple
mentation of P2 activities toward
health care industry' waste generation.
Successful P2 activities shall be recog
nized by awards or other recognition
by U.S. EPA, AHA and/or the Parties
acting jointly.
6.0 AGREEMENTS.
In order to foster the successful com
pletion of this MOU, the Parties agree
to the following terms and conditions:
4. Nothing in this MOU shall be con
strued to authorize or permit any vio
lation of any Federal, State or local
law imposed upon the Parties, includ
ing, but not limited to, the PRA,
APA, or the Anti-Deficiency Act, 31
U.S.C. 1342.
U .S .
10. AHA Environmental Leadership
Council. AHA will develop an AHA
Environmental Leadership Council
(“the Council”) that will be responsi
ble for making recommendations to
the AHA on educational and out
reach activities, recommending con
tent experts to participate in programs and/or the development of
products such as the Model Plans,
monitoring progress toward estab
lished environmental goals, selecting
the award recipients for national
recognition programs, and assisting in
the publication of an annual report
documenting the hospital industry’s
progress toward P2.
5.0 FUNDING. The Parties shall
attempt to secure reasonable funding
to allow for the successful completion
of the activities described herein. Both
Parties, however, expressly acknowl
edge that the activities under this
MOU shall be subject to the availabili
ty' of appropriated funds and personnel
of each Party, or the approval of other
sources of funding. Nothing in this
MOU or elsewhere shall be construed
as establishing a contract (or other
legally binding commitment) obligat
ing U.S. EPA or AHA to provide
money, goods or services of any kind
to any legal entity7.
any other written part of this MOU
must be made in writing and signed by
both Parties or their designees.
&
9. Industry Input on U.S. EPA
Guidance. To the extent feasible and
practical, U.S. EPA will solicit com
ments by AHA and the AHA
Environmental Leadership Council (as
established pursuant to this MOU) on
U.S. EPA’s policies and technical guid
ance specifically affecting the health
care industry’s waste streams. AHA’s
comments will be limited to the practi
cality7 and feasibility7 of the matters set
forth in the policies and technical guid
ance. Such input shall not be sought
with respect to any adjudication or any
rulemaking that is subject to the notice
and comment requirements set forth in
khe Administrative Procedure Act
(“APA”) at 5 U.S.C. 553(b).
these stakeholders play an important
role in the partnership to advance P2
in the health care industry'. In recogni
tion of this fact, the Parties will allow
for the participation of stakeholders in
the manner set forth in Attachment
#1 to this MOU.
A S S O C IA T IO N
work together to investigate P2 oppor
tunities with respect to ethylene oxide
and PBT pollutants.
nical assistance to accomplish the goals
of the MOU, U.S. EPA expressly
reserves the right to abstain from
expressing a position, either formal or
informal, on any matter of law, policy
or science related in any way to the
subject matter of this MOU, including,
but not limited to, any matter of law,
policy or science related to any PBT
pollutant. Nothing in this MOU shall
constitute any commitment by U.S.
EPA to investigate or reinvestigate any
position, either formal or informal on
any matter of law, policy or science.
8.0 TERMINATION.
Unless extended by a written agree
ment executed by both Parties, this
MOU shall terminate exactly five (5)
years from the date upon which this
MOU becomes fully executed by all
signatories listed below.
10. AHA shall maintain full right, title
and interest in any intellectual proper
ty right, including a copyright, in any
work product developed solely by
AHA under this MOU. Intellectual
property developed by AHA with
financial assistance from U.S. EPA
shall be subject to theconditions set
forth in U.S. EPA’s applicable assis
tance regulations (e.g., 40 C.F.R.
30.36). Any intellectual property
developed collaboratively by the
Parties will also be governed by the
Federal Copyright Statute at Title 17
of the United States Code or by rhe
Federal Patent Statute at Title 35 of
the United States Code.
For the United States Environmental
Protection Agency:
The Parties, on this 24th day of June,
1998, hereby agree to the foregoing
MOU, which shall be effective immedi
ately upon full execution by the signa
tories listed below.
Dr. William H. Sanders, III, Director
Office of Pollution Prevention and Toxics
Office of Prevention, Pesticides
and Toxic Substances
U.S. EPA
David A. Ullrich
Acting Regional Administrator
U.S. EPA, Region 5
For the American Hospital
Association:
Jonathan T. Lord, M.D.
Chief Operating Officer
American Hospital Association
11. Information on source reduction
received by U.S. EPA pursuant to this
MOU shall be made available to the
public pursuant to Section 6606(b) of
the PPA, 42 U.S.C. 13105(b).
7.0 PRIMARY CONTACTS.
The Parties intend that the work under
this MOU shall be carried out in the
most efficient manner possible. To that
end, the Parties intend to designate
individuals that will serve as primary
contacts between the Parties. The
Parties intend that, to the maximum
extent possible and unless otherwise
approved by the other Party, all signifi
cant communications between the
Parties shall be made through the pri
mary contacts. The designated primary'
contacts for the Parties are listed in
Attachment #2 to this MOU.
HeaLthfCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234-0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
Die IV F certification mark and term are the sole property of the Chlorine Free
Product* Association and arc only used by authorued and certified users.
Alternatives*
to Polyvinyl
Chloride (PVC)
and
Di-2-Ethylhexyl
Phthalate
(DEHP)
Medical
•Devices
Products detailed in this
publication include:
Ambulatory Products
Gloves, Examination
Intravenous (IV) products:
■ administration sets
0 bags
0 infusion tubes
Bedding Products
Patient ID Bracelets
Blood bags:
n fresh frozen plasma
a packed red blood cells
0 platelets
0 platelet rich plasma
Respirator)’ Therapy Products
0 endotracheal tubes
■ masks, aerosol and oxygen
0 oxygen hood
0 tracheostomy tubes
Body Bags
Sequential Compression Devices
Central line catheters and PICC lines
0 introcan safety catheters
0 midline catheters
0 percutaneous catheter introducers
0 peripherally-inserted central
catheters (PICC)
Total parental nutrition
0 bags
■ catheters
0 tubing
Umbilical vessel catheters
Dialysis, peritoneal
n rigid dialysate containers
0 peritoneal catheters
Enteral feeding sets
0 bags and tubing
0 extension sets
Enteral feeding nasogastric tubes
0 PEG tubes
0 gastrostomy tubes
0 nasoenteric tubes
0 nasogastic tubes
0 nasojejunal tubes
■ pediatric clear straight catheters
Urinary' drainage catheters
0 Foley catheters
■ urethral catheters for pediatrics
0 urinary catheters
Wound Drains and Drainage Systems
0 drains
0 nephrostomy catheters
■ surgical and wound drains
0 thoracic catheters
Office Supplies
Shower Curtains
Epidural vessel catheters
* Health Care Without Harm does not endorse any of these products, has not tested them
for safety or efficacy, and does not take responsibility' for the accuracy of the information
or product performance. Listing here is based solely on information provided by the man
ufacturer. Non-PVC products may contain much smaller amounts of DEHP Flexible
PVC-free products still must be tested to ascertain whether they are in fact DEHP-free.
Products that contain latex and chlorine are excluded from this table: latex products
because of concerns over latex allergies and chlorine containing products because of con
cerns over lifecycle hazards. Exceptions are made for the few PVC products for which few
or no non-PVC products are available. In those cases non-DEHP products are identified.
This table is a work-in-progress.
Sources: Sustainable Hospitals Project, 2000, “Alternative Products,” see http://sustainablehospitals.org (Lowell: Sustainable Hospitals Project, UMass Lowell); and Tickner, Joel,
et al, 1999, The Use of Di-2-Ethylhexyl Phthalate in PVC Medical Devices: Exposure,
Toxicity, and Alternatives (Lowell: Lowell Center for Sustainable Production, UMass
Lowell); and all information was verified through telephone contacts with manufacturer
representatives or review of manufacturer website information.
ALTERNATIVES
TO
PVC
AND
DEHP
MEDICAL
DEVICES
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 1 of 4)
Products
Manufacturer
Telephone
Webpage
Material
Comments
Ambulatory Products
Many manufacturers including
Merry Walker Corp.
815-678-3388
www.merrywalker.com
Steel
Product: Merry Walker
Bedding Products
Precision Dynamics Corp.
800-847-0670
www.pdcorp.com
Polyethylene
Disposable mattress and pillow
covers, draw sheets
Blood Bags
Baxter Healthcare, Fenwal Division
800-766-1077
www.baxter.com
Polyolefin
Bags for platelets, platelet rich plas
ma and fresh frozen plasma
Bags for packed red blood cells
Non-DEHP PVC
Body Bags
LASAN Plastics, Inc.
207-693-4817
www.lasan.com
Polyethylene/polypropylene blend
Central Line Catheters
and PICC Lines
B. Braun
800-227-2862
www.bbraunusa.com
Polyurethane or Teflon
Teflon or polyurethane
Percutaneous catheter introducers
Central venous catheter, introcan
safety catheter
•
Becton Dickinson
201-847-6800
www.bd.com
Silicone or polyurethane
Peripherally-inserted central
catheter, midLine catheter
Klein-Baker Medical
210-696-4061
www.neocare.com
Silicone
Peripherally-inserted central
catheter (neonates)
Utah Medical Products, Inc.
800-533-4984
www.utahmed.com
Silicone
Peripherally-inserted central
catheter (neonates)
Vygon
800-544-4907
www.vygonusa.com
Polyurethane or Silicone
Peripherally-inserted catheter
(adults and neonates)
Midline catheters (pediatrics or
adults)
Polyurethane
Dialysis, Peritoneal
Enteral Feeding Sets
B. Braun
800-621-0445
www.bbraunusa.com
Polypropylene/polyethylene comonomer
Rigid peritoneal dialysate container
Degania Silicone
401-658-0130
www.deganiasilicone.com
Silicone
Peritoneal catheter
Children's Medical Ventures
800-377-3449
www.childmed.com
Non-DEHP PVC
Enteral set
CORPAK MedSystems
800-323-6305
www.corpakmedsystems.com
Multi-layer bag: nylon, ethylene vinyl
acetate, polypropylene
Non-DEHP PVC
Non-PVC bag
Non-DEHP tube
Kendall Healthcare
800-962-9888
www.kendallhq.com
Non-DEHP PVC
Non-DEHP bag & tube
Vygon
800-544-4907
www.vygonusa.com
Polyethylene
Extension set tubes
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 2 of 4)
Products
Manufacturer
Telephone
Webpage
Material
Comments
Enteral FeedingNasogastric (NG)
Tubes
CORPAK MedSystems
800-323-6305
www.corpakmedsystems.com
Silicone
Polyurethane
Gastotrostomy tube for neonates
PEG tube for neonates, nasoenteric
feeding tube
C. R. Bard, Inc.
800-545-0890
•
www.bardmedical.com
Silicone
Polyurethane
Nasogastric tube for neonates
Pediatric clear staright catheter
Kendall Healthcare
800-962-9888
www.kendallhq.com
Polyurethane
Nasogastric tube, PEG feeding tube
Kimberly-Clark
(Ballard Medical Devices)
800-524-3557
www.kchealthcare.com
Silicone
PEG feeding tube, gastrotomy feed
ing tube, jejunal feeding tube
Klein-Baker Medical
210-696-4061
www.neocare.com
Silicone
Feeding tube for neonates
Ross
800-231-3330
www.ross.com
Polyurethane
Nasoenteric feeding tube, nasojejunal feeding tube
Gastrostomy tube (some peds), PEG
tube
Silicone
Epidural Vessel
Catheters
Gloves, Examination
Utah Medical Products, Inc.
800-533-4984
www.utahmed.com
Silicone
Nasogastric and nasojejunal tubes
(neonates/peds)
Vygon
800-544-4907
www.vygonusa.com
Polyurethane
Silicone
Gastric feeding tubes for infants,
sump tube (Salem or Replogal)
Nasojejunal tubes
Zevex
800-970-2337
www.zevex.com
Polyurethane
Nasoenteric feeding tube
B. Braun
800-227-2862
www.bbraunusa.com
Polyamide (Nylon)
Epidural vessel catheter
Vygon
800-544-4907
www.vygonusa.com
Polyethylene, polyurethane or
polyamide (nylon)
Epidural vessel catheter
Allegiance Healthcare Corp.
800-964-4227
www.allegiance.net
Nitrile
Ansell-Perry
800-321-9752
www.ansellhealthcare.com
Nitrile
Best Manufacturing Co.
800-241-0323
www.bestglove.com
Nitrile
ECI Medical Technologies
902-543-6655
www.ecimedical.com
Styrene butadiene
Maxxim Medical
800-727-7951
www.maxximmedical.com
Polyurethane
S 3
I A 3 a
1 V 3 I a 3 W
d H 3 Q
0 N V
3 A d
01
S 3 A I 1 V N d 3 1 3 V
ALTERNATIVES
TO
PVC
AND
DEHP
MEDICAL
DEVICES
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 3 of 3)
Products
Manufacturer
Telephone
Webpage
Material
Gloves, Examination
(continued)
Safeskin Corporation
800-462-9993
www.safeskin.com
Nitrile
SmartCare Inc.
800-822-8956
www.smartcare.com
Nitrile
Tillotson Healthcare Corp.
800-445-6830
www.thcnet.com
Nitrile
B. Braun
800-227-2862
www.bbraunusa.com
Multi-layer bag: Polypropylene/polyethylene copolymer, polyester, elastomer
laminate
Polypropylene/polyethylene copolymer
Polyethylene
IV bag (Excel)
Intravenous (IV) Bags
and Tubing
Comments
IV bag (PAB)
IV set with PVC-free tube (no
longer manufacturing, but still
available from some vendors)
Budget Medical Products
800-569-1620
www.icumed.com
Non-DEHP PVC
IV tube
Children's Medical Ventures
800-377-3449
www.childmed.com
Non-DEHP PVC
IV administration sets
Curlin Medical
714-893-2200
www.curlinmedical.com
Non-DEHP PVC
Infusion tube
Office Supplies:
3-ring binders
Available from standard office
supply companies
800-847-0670
Patient ID Bracelets
Precision Dynamics Corp.
800-521-5123
www.pdcorp.com
Tyvek®
TabBand
800-940-3993
www.tabband.com
Tyvek®, polypropylene and polyethylene
Wristband & Medical Specialty
Products
800-348-6064
www.wristbandsupply.com
Tyvek®
Appropriate for short stays
Bivona Medical Technologies
800-847-8000
www.bivona.com
Silicone
Endotracheal tube, tracheostomy
tube
DHD Healthcare
800-553-5214
www.dhd.com
Silicone
Aerosol mask
Rusch
800-533-4984
www.ruschinc.com
Red rubber or silicone
Reusable endotracheal tube
Utah Medical Products, Inc.
800-932-0760
www.utahmed.com
Co-polyester--polyethylene foam and
polypropylene
Disposable infant oxygen hood
Vital Signs
800-962-9888
www.vital-signs.com
Polyester
Oxygen or aerosol applicationsAero2Mask
Respiratory Therapy
Products
Polyethylene, cardboard
Appropriate for short stays
Alternatives to Polyvinyl Chloride (PVC) and Di-2-Ethylhexyl Phthalate (DEHP) Medical Devices (Part 4 of 4)
Products
Manufacturer
Telephone
Webpage
Material
Sequential
Compression Device
Kendall Healthcare
800-846-3000
www.kendallhq.com
Polyolefins
Shower Curtains
Brookstone
800-222-6883
www.brookstone.com
Tyvek®
Many manufacturers
Total Parenteral
Nutrition
Umbilical Vessel
Catheters
Urinary Catheters
Wound
Drains/Drain age
Systems
Comments
Nylon
Abbott
800-766-1077
www.abbott.com
Non-DEHP PVC
Empty IV bag and tube
Baxter Healthcare, Fenwal Division
800-544-4907
www.baxter.com
Ethylene vinyl acetate
TPN bag
Vygon
800-962-9888
www.vygonusa.com
Polyurethane
Catheter for parenteral nutrition
and mid/long-term IV therapy
(See PICC lines above)
Kendall Healthcare
210-696-4061
www.kendallhq.com
Polyurethane
Umbilical vessel catheter
Klein-Baker Medical
800-533-4984
www.neocare.com
Silicone
Umbilical vessel catheter
(neonates)
Utah Medical Products, Inc.
800-544-4907
www.utahmed.com
Silicone or polyurethane
Umbilical vessel catheter
Vygon
800-545-0890
www.vygonusa.com
Polyurethane
Umbilical vessel catheter
C.R. Bard
800-658-0130
www.bardmedical.com
Polyurethane
Urethral catheter for pediatrics
Degania Silicone
210-696-4061
www.deganiasilicone.com
Silicone
Foley catheter
Klein-Baker Medical
800-533-4984
www.neocare.com
Silicone
Urinary drainage catheter
(neonates)
Utah Medical Products, Inc.
800-545-0890
www.utahmed.com
Silicone
Urinary catheters
C.R. Bard
401-658-0130
www.bardmedical.com
Silicone
Drains
Degania Silicone
800-533-4984
www.deganiasilicone.com
Silicone
Surgical and wound drains, tho
racic catheter, nephrostomy
catheter (may fit neonates)
www.utahmed.com
Silicone
Thoracic catheter
Utah Medical Products, Inc.
S 2
I A 3 a
1 V 3 I a 3 W
dH3a
0 N V
3 A d
01
S3AI1VNM311V
✓
i
I
I
Health Care Without Harm does not endorse any of these products, has not test
ed them for safety or efficacy, and does not take responsibility for the accuracy of
the information or product performance. Listing here is based solely on informa
tion provided by the manufacturer. Non-PVC products may contain much smaller
amounts of DEHP Flexible PVC-free products still must be tested to ascertain
whether they are in fact DEHP-free. Products that contain latex and chlorine are
excluded from this table: latex products because of concerns over latex allergies
and chlorine containing products because of concerns over lifecycle hazards.
Exceptions are made for the few PVC products for which few or no non-PVC
products are available. In those cases non-DEHP products are identified. This
table is a work-in-progress.
a
Sources: Sustainable Hospitals Project, 2000, “Alternative Products,” see
http://sustainablehospitals.org (Lowell: Sustainable Hospitals Project, UMass
Lowell); and Tickner, Joel, et al, 1999, The Use of Di-2-Ethylhexyl Phthalate in
PVC Medical Devices: Exposure, Toxicity, and Alternatives (Lowell; Lowell
Center for Sustainable Production, UMass Lowell); and all information was veri
fied through telephone contacts with manufacturer representatives or review of
manufacturer website information.
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 2O2.234.OO91
Fax: 202.234.9121
www.nohann.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at wvAV.noharm.org.
The PCF certification mark and term air the
ph'pvnx »•! the Chk’nnc Free
Products Association and arc onh med h authorized and ccrtilied incr<.
List of
Resolutions on
PVC, Dioxins,
Mercury
Resolution or Ordinance
Date
Issues Covered
American Medical Association
March 2001
Mercury, Lead,
Benzene
American Medical Women's Association
Nov. 1999
PVC, Dioxin,
Incineration
American Nurses Association
1997
PVC, Incineration,
Mercury
American Public Health Association
Nov. 1996
PVC, Dioxin
Association of Bay Area Governments
Sep. 1999
PVC, Dioxin
California Medical Association
March 2001
DEHP
California Medical Association
Feb. 199S
PVC, Dioxin
California Medical Association
March 2000
PVC, Dioxin,
Incineration
California Medical Association
March 2000
Mercury
Catholic Health Association or Minnesota
April 1999
PVC, Dioxin
Chicago, Illinois City Council
July 2001
Mercury
Chicago Medical Society
1998
PVC, Dioxin
Chicago Medical Society
Oct. 2001
Mercury
Chicago Medical Society
Oct. 2001
DEHP in NICUs
Church of the Brethren General Board
Oct. 1996
PVC, Dioxin
City and Count}' of San Francisco, CA
Sep. 199S
PVC, Dioxin
City and County of San Francisco
Commission on the Environment
March 1999
PVC, Dioxin
City Council of Ann Arbor, Michigan
July 2000
Mercury
City Council of Duluth, Minnesota
March 2000
Mercury
City of Berkeley, California
Oct. 199S
PVC, Dioxin
City of Berkeley, California
Oct. 2000
PVC, Dioxin
City of Boston, Massachusetts
Nov. 2000
Mercury
City of Los Angeles, California
Aug. 2000
Mercury
City of San Francisco, California
Feb.2000
Mercury
Cohasset, Massachusetts City Council
March 2001
Mercury
Common Council of Stoughton, Wisconsin
Oct. 2000
Mercury
Council of Dane County, Wisconsin
July 2000
Mercury
County of Santa Clara Office of
the Board of Supervisors
June 1998
PVC, Dioxin
Fergus Falls. Minnesota City Council
Dec. 2000
Mercury
Haverhill, Massachusetts City Council
March 2C01
Mercury
International Council of Nurses
1998
PVC, Dioxin,
Latex, Mercury,
Incineration
International Society of Doctors
for the Environment
Oct. 1999
PVC, Dioxin
Marin County Board of Supervisors
Dec. 1999
PVC, Dioxin
Massachusetts Medical Society
Nov. 2000
Mercury
Minnesota Academy of Family Physicians
April 1999
PVC, Dioxin
Minnesota Health and Housing Alliance
Dec. 1999
PVC, Dioxin
Minnesota Medical Association
House of Delegates
Oct. 1998
PVC, Dioxin
Minnesota Public Health Association
1996
PVC, Dioxin
Natick, Massachusetts City Council
April 2000
Mercury
Oakland City Council
Feb. 1999
PVC, Dioxin
Racine, Wisconsin Common Council
March 2001
Mercury
Religious Action Center of Reform Judaism April 1999
PVC, Dioxin,
Incineration,
Mercurv
State of Indiana
Mav• 2001
Mercury
State of Maine
June 2001
Mercury
State of Maryland
May 2001
Mercury'
State of Minnesota
April 2001
Mercury
State of New Hampshire
June 2000
Mercury
State of Oregon
Aug. 2001
Mercury
State of Rhode Island
July 2001
Mercury
Tenet Healthcare Corporation
Oct. 1999
PVC, Dioxin
United Methodist Church
April 1996
PVC, Dioxin
United Methodist Church
2000
PVC, Dioxin
Universal Health Services
May 1999
PVC, Dioxin
Village Board of DeForest, Wisconsin
Sept. 2000
Mercury
Wisconsin Public Health Association
June 2001
Mercury
Worcester, Massachusetts City Council
May 2001
Mercury
HealthrCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20C09
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit or to find out how to get a complete kit.
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF ccraikacion rrurk jo] term arc the wk pnf’crty of chc Chlonnc Free
Pnxiucu Auccutmn and arc only used by authored and ccruricd uxn.
on PVC and
DEHP in
Health Care
US Government Publications
Other Materials on PVC or DEHP
in Healthcare
U.S. Food and Drug Administration
(FDA), Center for Devices and
Radiological Health. 2001. Safety
Health Care Without Harm. 2001.
Assessment of Di(2-ethylhexyl) Phthalate
(DEHP) Released from PVC Medical
Devices. Rockville, MD: U.S. FDA.
Washington, DC; Health Care Without
Harm. Webpage: www.noharm.org.
Dioxin, PVC and Health Care.
Health Care Without Harm. 2001.
National Toxicology Program, Center for
the Evaluation of Risks to Human
Reproduction (CERHR). 2000. NTP
CERHR Expert Panel Report on
Di (2-ethylhexyl) Phthalate. Webpage:
http://cerhr.mehs.nih.gov/newsAndex.html
United State Environmental Protection
Agency (US EPA). 2000. Draft Exposure
and Human Health Reassessment of
2,3,7,8'TetrachlorodibenzO'p'Dioxin
(TCDD) and Related Compounds.
Washington, DC: US EPA. Webpage:
http://www.epa.gov/ncea/pdfs/dioxin/part
land2.htm.
European Government
Publications
Swedish National Chemicals
Inspectorate. 2000. Risk. Assessment:
bis (2-ethylhexyl) phthalate (Final Draft).
Soina, Sweden.
Danish Ministry of Environment and
Energy. 1999. Action Plan for Reducing
and Phasing Out Phthalates in Soft Plastics.
Copenhagen, Denmark.
European Commission. 2000. Green
Paper on Environmental Issues of PVC.
Webpage: www.europa.eu.int/
comm/environment/pvc/index.htm
European Commission. 2000.
Five PVC studies:
1. The Influence of PVC on the Quantity
*
and Hazardousness of Flue Gas
Residues from Incineration
2. Economic Evaluation of PVC Waste
Management
3. The Behaviour of PVC in Landfill
4. Chemical Recycling of Plastics Waste
(PVC and Other Resins)
5. Mechanical Recycling of PVC Wastes
Webpage: www.europa.eu.int/comm/
environment/waste/facts en.htm
Reducing PVC Use in Hospitals.
Washington, DC: Health Care Without
Harm. Webpage: www.noharm.org.
Health Care Without Harm. 2001. A
Summary of the FDA Safety Assessment of
DEHP Released from PVC Medical
Devices. Washington, DC: Health Care
Without Harm. Webpage:
www.nohamr.org.
Neonatal Exposure to
DEHP and Opportunities for Prevention.
Rossi M. 2000.
Falls Church, VA: Health Care Without
Harm. Webpage: www.noharm.org.
Rossi M, Schertler T. 2000. “PVC
White Paper." In Proceedings from Setting
Healthcare's Environmental Agenda (San
Francisco, CA). Falls Church, VA:
Health Care Without Harm.
Schertler T. 1999. Do We Have a Right
to Higher Standards? C. Everett Koop, MD
and an ACSH Panel Review the Toxicity
and Metabolism of DEHR Falls Church,
VA: Health Care Without Harm.
Webpage: www.noharm.org.
Schertler T. 2001. DEHP Exposures
During the Medical Care of Infants: A
Cause for Concern. Washington, DC:
Health Care Without Harm. Webpage:
www.noharm.org.
Silas J. 2001. A Summary of the Expert
Panel Report of the National Toxicology
Program on DEHP and its Risks to Human
Reproduction. Washington, DC: Health
Care Without Harm. Webpage:
www.noharm.org.
Tickner J, Schertler T, Guidotti T McCally
M. Rossi M. 2001. “Health Risks Posed by
Use of Di-2-Ethylhexyl Phthalate (DEHP)
in PVC Medical Devices: A Critical
Renew.” American Journal of Industrial
Medicine, 39:100-111.
Tickner J, Hunt F} Rossi M, Haiama N,
Lappe M. 1999. The Use of Di-2-
Ethylhexyl Phthalate in PVC Medical
Devices: Exposure, Toxicity, and
Alternatives. Lowell: Lowell Center for
Sustainable Production, University of
Massachusetts Lowell. Webpage:
www.noharm.org.
University of Massachusetts Lowell,
Sustainable Hospitals Project. 2000.
“Alternative Products.” Webpage:
www.sustainablehospitals.org.
Video: “First Do No Harm: PVC and
Medicine’s Responsibility.” Western
Lake Superior Sanitary District, MN.
(2000). (For copies contact Health Care
Without Harm at hcwh@chej.org or
202-234-0091).
Without Harm
1755 S Street. NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Gwn: A Resource Kit for Pollution
Prevention in Health Core. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF ccrutkataon mark irul terni Are the *o!e property of the Chicane Free
Product! Allocution and are enS u>eJ b* audkxucd and ceniFxd uierv
Resources
on PVC and
DEHP in
Health Care
US Government Publications
Other Materials on PVC or DEHP
in Healthcare
U.S. Food and Drug Administration
(FDA), Center for Devices and
Radiological Health. 2001. Safety
Health Care Without Harm. 2001.
Assessment of Di (2-echylhexyl) Phthalace
(DEHP) Released from PVC Medical
Devices. Rockville, MD: U.S. FDA.
Washington, DC; Health Care Without
Harm. Webpage: www.noharm.org.
Dioxin, PVC and Health Care.
Health Care Without Harm. 2001.
National Toxicology Program, Center for
the Evaluation of Risks to Human
Reproduction (CERHR). 2000. NTP
CERHR Expert Panel Report on
Di (2'ethylhexyl) Phthalate. Webpage:
http://cerhr.niehs.nih.gov/news/index.html
United State Environmental Protection
Agency (US EPA). 2000. Draft Exposure
and Human Health Reassessment of
2,3.7,8'TetrachlowdibenzO'p'Dioxin
(TCDD) and Related Compounds.
Washington, DC: US EPA. Webpage:
http://www.epa.gov/ncea/pdfs/dioxin/part
land2.htm.
European Government
Publications
Swedish National Chemicals
Inspectorate. 2000. Risk Assessment.
bis (2-ethylhexyl) phthalate (Final Draft).
Soina, Sweden.
Danish Ministry of Environment and
Energy. 1999. Action Plan for Reducing
and Phasing Out Phthalates in Soft Plastics.
Copenhagen, Denmark.
European Commission. 2000. Green
Paper on Environmental Issues of PVC.
Webpage: www.europa.eu.int/
comm/environment/pvc/index.htm
European Commission. 2000.
Five PVC studies:
1. The Influence of PVC on the Quantity
and Hazardousness of Flue Gas
Residues from Incineration
2. Economic Evaluation of PVC Waste
Management
3. The Beliaviour of PVC in Landfill
4. Chemical Recycling of Plastics Waste
(PVC and Other Resins)
5. Mechanical Recycling of PVC Wastes
Webpage: www.europa.eu.inc/comm/
environment/waste/facts_en.htm
Reducing PVC Use in Hospitals.
Washington, DC: Health Care Without
Harm. Webpage: www.noharm.org.
Health Care Without Harm. 2001. A
Summary of the FDA Safety Assessment of
DEHP Released from PVC Medical
Devices. Washington, DC: Health Care
Without Harm. Webpage:
www.noharm.org.
Rossi M. 2000. Neonatal Exposure to
DEHP and Opportunities for Prevention.
Falls Church. VA: Health Care Without
Harm. Webpage: wwxv.noharm.org.
Rossi M, Schertler T. 2000. "PVC
White Paper." In Proceedings from Setting
Healthcare's Environmental Agenda (San
Francisco, CA). Falls Church, VA:
Health Care Without Harm.
Schertler T. 1999. Do We Have a Right
to Higher Standards? C. Everett Koop. MD
and an ACSH Panel Review the Toxicity
and Metabolism of DEHR Falls Church,
VA: Health Care Without Harm.
Webpage: www.noharm.org.
Schertler T. 2001. DEHP Exposures
During the Medical Care of Infants: A
Cause for Concern. Washington, DC:
Health Care Without Harm. Webpage:
www.noharm.org.
Silas J. 2001. A Summary of the Expert
Panel Report of the National Toxicology
Program on DEHP and its Risks to Human
Reproduction. Washington, DC: Health
Care Without Harm. Webpage:
www.noharm.org.
Tickner J, Schertler T, Guidotti T McCally
M, Rossi M. 2001. “Health Risks Posed by
Use of Di-2-EthylhexyI Phthalate (DEHP)
in PVC Medical Devices: A Critical
Review.” American Journal of Industrial
Medicine, 39:100-111.
Tickner J, Hunt P, Rossi M, Haiama N,
Lappe M. 1999. The Use of Di-2 ~
Ethylhexyl Phthalate in PVC Medical
Devices: Exposure, Toxicity, and
Alternatives. Lowell: Lowell Center for
Sustainable Production, University of
Massachusetts Lowell. Webpage:
www.noharm.org.
University of Massachusetts Lowell,
Sustainable Hospitals Project. 2000.
“Alternative Products." Webpage:
www.sustainablehospitals.org.
Video: “First Do No Harm: PVC and
Medicine’s Responsibility." Western
Lake Superior Sanitary District, MN.
(2000). (For copies contact Health Care
Without Harm at hcwh@chej.org or
202-234-0091).
HealthrCare^
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF ccruncitnn mark and term arc the wlc property ui* the Chkw.c Few
Prcxiucu Amccucsog and are only used by auchorued and certified uxn.
Problem
Fast Facts
n
Mercury is a neurotoxic, heavy
metal that is linked to numerous
health effects in wildlife and people.
°
Mercury can be found throughout
hospitals in products such as ther
mometers, sphygmomanometers,
dilation and feeding tubes, batter
ies, fluorescent lamps, thermostats,
and bleach.
°
The most likely routes of exposure
arc inhalation of inorganic mercu
ry vapor after a spill or during a
manufacturing process, or inges
tion of methylmercury from con
taminated fish.
o
Mercury can pose a significant
health threat when spilled in a
small, poorly ventilated room.
D
A report issued by the National
Academy of Sciences National
Research Council estimated that
every year 60,000 children are at
risk of being born in the United
States with neurological problems
that could lead to poor school per
formance because of exposure to
methylmercury in uteroJ
■
A study by the Centers for Disease
Control estimated that 1 in 10
women currently have mercury lev
els in their bodies high enough to
cause neurological effects in their
offspring."
□
There is approximately 1 gram of
mercury in a typical fever ther
mometer. This is enough mercury
to contaminate a lake with a sur
face area of about 20 acres, to the
degree that fish would be unsafe to
eat.'"
■
There are over 1,900 fish advisories
in place on water bodies across the
U.S. due to mercury contamina
tion.
°
In March 2001, the FDA released a
consumer advisory that warned
pregnant women not to eat shark,
swordfish, king mackerel or tilefish,
because they contain enough mer
cury to damage the fetus’s nervous
system. Young children, nursing
mothers and women who may
become pregnant were advised to
avoid those fish as well.*
°
Hospitals contribute 4-5% of the
total wastewater mercury1 load.'
■ There is up to 50 times more mer
cury in medical waste than in gen
eral municipal waste, and the
amount of mercury emitted from
general medical waste incinerators
averages more than 60 times that
from pathological incinerators.'1
□
Medical and solid waste that con
tains mercury' or has been contami
nated by mercury' is considered haz
ardous waste and should be kept
out of the waste stream.
a
In 2000, the mercury from fever
thermometers accounted for 17
tons or 10% of mercury' in the
municipal solid waste stream.'"
°
In 2000, mercury from batteries
made up 98 tons or 57% of mercury'
in the municipal solid waste
stream.'1"
■
Legislation banning the sale of mer
cury' thermometers has been passed
in the cities of Duluth, MN; Ann
Arbor, MI; San Francisco, CA
Boston, MA; Chicago, IL; and in
the states of Maryland, Maine,
Minnesota and New Hampshire.
Legislation is pending in a host of
other cities, stares and in Congress.
Notes
i.
National Academy of Sciences National
Research Council. July 2000. “Toxicological
Effects ot Methylmercurv."
•
•
ii.
CDC Morbidity and Mortality Weekly
Report- "Blood and Hair Mercury Levels in
Young Children and Women of Child
Bearing Age-United States.” 1999 Vol 50,
No 08.140. 03/02/2001
iii. Personal Communication. Jamie Harvie. PE.
Institute for a Sustainable Future. 218-5257806.
iv
U.S. Food And Drug Administration. Center
for Food Safety and Applied Nutrition.
www.cfsan.fda.gov March 2001.
v.
Personal Communication. Western Lake
Superior Sanitary District, Duluth. MN.
vi. USEPA. Mercury in Medical Waste: Keeping
Mercury out of Medical Waste.
www.epa.gov/reg5oair/glakes fact l.htm
vn USEPA. Background Information on
Mercury Sources and Regulations.
www. e p a. gov/g r 11 a kes/b nsdoc s/ mere srce/merc_srce.html#Table 2B
viii. USEPA. Background Information on
Mercury Sources and Regulations.
www.epa.gov/grtlakes/bnsdocs/mercsrce/
merc_srce html#Table 2B
HealthrCare
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care, For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
I^IsoyinkI.
The PCF certification mark and term arc the %o!c property of the Chlorine Free
Product) /Xvoctatfoti and arc only u*ed by authorized and certified u -er*.
Making
Medicine
Mercury-Free
A Resource Guide
for Mercury-Free
Medicine
Mercury Elimination—
Preventive Medicine
for Human Health and
The Environment
Health Care Without Harm (HCWH)
is an international coalition of more
than 340 organizations in 37 countries,
working together to eliminate pollution
from health care practices without
compromising safety or care. Health
Care Without Harm is committed to
transforming the health care industry
so that it is no longer a source of envi
ronmental harm.
Why Mercury has No
Business in the Health
Care Business
Mercury can be found in many health
care devices, including fever ther
mometers, blood pressure cuffs, and
esophageal dilators. Mercury is also
found in many chemicals and measure
ment devices used in health care labo
ratories. If these products are spilled,
broken or disposed of improperly, there
is a potential for significant harm to
human health and the environment.
Medical waste incinerators, as well as
municipal waste incinerators, emit
mercury when they burn wastes that
contain mercury. According to the
U.S. Environmental Protection Agency
(EPA), medical waste incinerators are
the fourth largest source of mercury to
the environment.1 Hospitals are also
known to contribute 4'5% of the total
waste water mercury load.2
Mercury-containing devices improperly
disposed of in a landfill are also a
potential source of harm. Mercury
fever thermometers alone contribute
about 17 tons of mercury to solid waste
landfills annually.1
Health Implications
Mercury is a reproductive toxin and a
potent neurotoxin—it affects the brain
and the central nervous system.
Pregnant women, women of child
bearing age and small children are at
the greatest risk. Mercury can cross the
placenta and cause irreparable neuro
logical damage to the fetus. A National
Academies of Science report from July,
2000 showed that 60,000 children are
bom in the United States each year
with neurological problems that, could
lead to poor school performance
because of exposure to methylmercury
in utero.4 In March, 2001 a study from
the Centers for Disease Control and
Prevention sampled the mercury levels
in the blood, hair and urine of women
and children and found that one in 10
women have mercury levels high
enough to cause their children neuro
logical damage — putting about
395,000 babies a year in danger.5
Environmental Implications
Mercury in the air is transported to
water bodies primarily through precipi
tation. Mercury released to the envi
ronment can cause early death, weight
loss, and reproductive problems in
wildlife. In fish, (methyl) mercury' can
concentrate to levels one million times
higher than those in the surrounding
water. Over forty' states have fish con
sumption advisories because of wide
spread mercury' contamination.
LU
Economic Implications
LU
LL_
I
or
o
or
LU
S
LU
21
►—«
*—«
o
LU
Many hospitals are now required to
meet strict wastewater treatment dis
charge limits for mercury', and this reg
ulatory trend is likely to continue.
Mercury spill training is costly and in
some parts of the country', JCAHO has
issued recommendations to hospitals
for inadequate staff training on mercu
ry clean-up. There are also countless
stories of mercury' spills in hospitals
where mercury' cleanup costs have
been substantially higher than the cost
of mercury-free alternatives. By elimi
nating mercury, hospitals can not only
protect the health of local communi
ties, but their “bottom line” as well.
o
2:
<
Regulatory Implications
By Federal law, mercury' is a regulated
waste; as a result, its management can
be quite expensive. Using mercurycontaining devices requires a “mercury'
management policy” and a spill
response plan for emergencies. Costs
associated with mercury spills are high
— often in the thousands ol dollars —
and can pose health risks for health
care staff and patients. Compliance
with regulations for disposal of mercu
ry-contaminated waste with a hazardous waste hauler may result in
incineration of the waste,.with subse
quent mercury emissions to the envi
ronment. In addition, elemental mer
cury' recycling may keep it out of the
environment in the short-term, but its
reuse in new products poses the same
eventual risk to human health.
Hospitals can avoid the risk of mercu
ry’ management altogether by using
non-mercury alternatives. As leading
health care institutions across the
country', such as Dartmouth-Hitchcock
Medical Center, the Mayo Clinic
and the National Institutes of Health
now recognize, safe and effective alterna
tives exist for nearly all traditional health
care uses of mercury’, from temperature
and blood pressure measurement to
fixatives used in the lab.
How Health Care
Without Harm is
Addressing the Problem
In 1998, Health Care Without Harm
kicked off the ‘Making Medicine
Mercury-Free’ program by asking hos
pitals to take a pledge to phase-out
mercury'-containing products in their
facilities. HCWH has also provided
educational and technical resources to
those hospitals implementing mercury
elimination programs. As of August,
2001, over 600 hospitals and clinics
had signed the pledge to go mercuryfree.
Cities and stales across the country are
taking a proactive approach to mercu
ry elimination. For example, San
Francisco, CA; Ann Arbor, MI;
Duluth, MN; Boston, MA; Chicago,
IL; and states, such as New
Flampshire, Maine, Maryland and
Minnesota have prohibited the sale,
manufacture and distribution of mer
cury' thermometers within their juris
dictions. City, state and federal legis
lation is pending across the country to
eliminate the use of mercury' and mercury-containing products.
What Your Hospital Can Do
In September, 2001 HCWH and the
Hospitals for a Healthy Environment
(H2E) partnership merged their pledge
programs into one national pledge ini
tiative. H2E is a partnership between
HCWH, the American Hospital
Association (AHA), the American
Nurses Association (ANA) and the
Environmental Protection Agency
(EPA). As part of this collaboration,
HCWH will continue to assist health
care providers in meeting this impor
tant mercury elimination goal.
What States and
Cities are Doing
Communities across the country are
holding mercury' thermometer
exchanges in an effort to get rhe toxic
metal out of family medicine cabinets
and to educate the community about
the dangers of mercury'. The
exchanges are designed so that resi
dents can bring in their mercury ther
mometers for recycling and receive a
new non-mercury alternative at the
same time. To assist in these efforts,
HCWH has developed a resource
guide entitled How to Plan and Hold a
Mercury Fever Thermometer Exchange.
You can order this and several other
publications or sign up for the H2E
listserv by filling out the form on page
four.
Take the Making Medicine
Mercury-Free Pledge and join the
hundreds of medical facilities
across the country' that have heguiw
the process of eliminating their use
of mercury'. The eventual elimina
tion of mercury-containing prod
ucts is the only way to keep mercu
ry out of the environment and to
reduce its impacts on human
health. To take the pledge, please
visit the website (www.h2eonline.org) and gain recognition
for your commitment to improving
environmental health. Once you
have embarked on your mercury
elimination program, you will be
eligible to receive a Making
Medicine Mercury' Free Award.
1 Join the H2E listserv to share and
learn technical information, find
educational tools and identify' prac^x
tical strategies for mercury' elimina
tion and discuss other pollution
prevention and waste minimization
issues. To join the listserv, go to
the HCWH website
(www.noharm.org) and click on
the H2E logo.
Conduct a mercury audit to iden
tify' all uses and sources of mercury
in your institution.
Commit to eliminate the use of
mercury' by investigating opportu
nities to phase out mercury-con
taining items where fewer barriers
exist and immediate steps can be
taken. For example, investigate
replacing mercury-filled patient
□
Eliminating Mercury Use in Hospital Laboratories:
A Step toward Zero Discharge: Public Health
Reports, July/August 1999 Volume 114 p353358.
Medical waste pollution prevention. Keep mercury
out of the wastewater stream. U.S. Environmental
Protection Agency, Region 5. Chicago, IL.
Mercury. Western Lake Superior Sanitary
District. Duluth, MN.
Mercury Pollution Prevention in Healthcare: A
Prescnption for Success. National Wildlife
Federation, Great Lakes Natural Resource
Center. NWF/Great Lakes Natural Resource
Center, 506 East Liberty, 2nd Floor, Ann Arbor
MI 48104- 2210. (734) 669-3351.
Mercury Use in Hospitals ami Clinics 20-minute
video and guidebook. Minnesota Office of
Environmental Assistance, 520 Lafayette Road
N„ 2nd Floor, St. Paul, MN 55155, (612) 2963417, (800) 657-3843.
Pollution Prevention for Hospitals and Medical
Facilities and Best Management Practices for
Hospitals and Medical Facilities Palo Alto
Regional Water Quality’ Control Plant, 2501
Embarcadero Way, Palo Alto CA 94303. (415)
329- 2598
Reducing Mercury Use in Health Care, Promoting a
Healthier Environment, A How-to Manual.
Monroe County, New York Department of
Health (716) 292-3935
The Case Against Mercury: R.x for Pollution
Prevention (poster and booklet). Created in
cooperation with U.S- Environmental Protection
Agency. Terrine Institute, 4 Herbert Street,
Alexandria VA 22305, Region 5.(703) 5485473 www.terrene.org.
Video
Resource List
Publications
How to Plan and Hold a Mercury Fever
Thermometer Exchange
Mercury Thermometers and Your Family’s Health
Making Medicine Mercury Free
The Mercury Problem- Fast Facts
What s Wrong With Mercury Thermometers?
Health Care Without Harm, 1755 S St.. NW
Suite 6B Washington, DC 20009
(202) 234-0091 or info®hcwh.org
Health Care Without Harm
xvww.noharm.org
Hospitals for a Healthy Environment (H2E)
www.h2e-online.org
First Do No Harm, Environmental Working
Group, 1997.
www.ewg.org/pub/home/HCWC/hcwh.html
Harvard University: Laboratory Thermometers
www.uos.harvard.edu/ehs/factsheets/
ea_mercury_therm.html
Indiana Department of Environmental
Management: Factsheet on Mercury
Thermometers
www.state.in.us/idcm/ctap/mercury/
lhermometers.html
Massachusetts Water Resources Authority'
www.mwra.state.ma.us
Massachusetts Medical, Academic and Scientific
Community Organization (MASCO)
www. m a s c o. o rg/m e rc u ry
Mercury’ in Medical Waste: Keeping Mercury'
Out of Medical Waste
www.p2pays.0rg/ref/01/00792. him
Mercury' Use Reduction & Waste Prevention in
Medical Facilities
Educational software for the Web by USEPA
Region 5 and Purdue University
www.cpa.gov/seahome/mercury/src/title.htm
Minnesota Office of Environmental Assistance.
Mercury for Health Professionals
www.moea.state.mn.us/res/V7_2/mercury.cfm
National Institutes of Health Mad as a
Hatter—Campaign for a Mercury-Free NIH
www. n i h. gov/o d/ors/ds/nome rc u ry/
Notes
1.
2.
3.
Agents of Change. Mercury Waste Solutions.
American Hospital Association:
1-800-AHA-2626
4.
Mercury and the Healthcare Professional (17 min
utes). Minnesota Office of Environmental
Assistance and the US EPA, Region 5. Contact
Emily Moore, Minnesota Office of
Environmental Assistance, 520 Lafayette Road
N., 2nd Floor, St. Paul MN,55155- 4100. (612)
215-0201.
5.
1997 Mercury' Report to Congress
Personal communication, Western Lake
Superior Sanitary District, Duluth, MN.
(U.S. Environmental Protection Agency,
1996, Mercury Study Report to Congress,
Science Advisory Board Review Draft, Vol.
2, p. 4-19, p. ES-3.)
CDC Morbidity and Mortality Weekly
Report. “Blood and Hair Mercury Levels in
Young Children and Women of ChildBearing Age—United State." 1999 Vol. 50
No. 08:140, 03/02/2001.
National Academies of Science, National
Research Council, July 2000. “Toxicological
Effects of Methylmercury.”
M E R C U R Y -F R
You may also contact the technical
staff listed on the H2E website
tfwww.h2e-online.org) for continued
technical assistance and support.
Blueprint for Mercury Elimination Western Lake
Superior Sanitary District; (38-page book of
interest 218-722-3336, free).
Web Resources
M E D IC IN E
For more information, please contact:
Health Care Without Harm
1755 S Street, NW, Suite 6B
Washington, DC 20009
www.noharm.org
Phone: 202-234'0091
Fax: 202-234'9121
E-mail: info@hcwh.org
Becoming a Mercury-Free Facility: A Priority to be
Achieved by the Year 2000. Hollic Shaner, RN
MSA. AHA/ASHES - 1-800-AHA-2626
K IN G
thermometers with digital or elec
tronic thermometers; replacing
mercury’ with water in Miller
Abbott Tubes, replacing mercurycontaining bougies or esophageal
dilators with silicon ones; or
replacing mercury-filled blood pres
sure measuring devices with
aneroid units.
■ Implement a “Mercury-Free
Purchasing Policy.” Assign materi
als management staff to communi
cate with suppliers about the policy
and to work with staff on finding
non-mercury alternatives.
o Educate and train your employ
ees about facility' protocols, includ
ing information about mercury and
its effects on human health and the
environment.
J Hold a mercury' thermometer
exchange for your employees.
■ Discontinue sending mercury
thermometers home with the par
ents of newborns and other
patients.
Making Medicine Mercury-Free
Contact Name: _______________________________________________________
Ti tie:
_____________________________________________________
I
Organization:
Address:
______________________________
_______________________________________________
City, State,Zip:
Phone: ()Fax: ()
I am interested in HCWH educational materials. Please send me the following
publications:
O How to Plan and Hold a Mercury’ Thermometer Exchange
O Mercury’ Thermometers and your Family’s Health
O Replacing Mercury’ Sphygmomanometers
O The Mercury’ Problem- Fast Facts
O Thermometer Fact Sheet
O Hospitals for a Healthy Environment (H2E) Information
O Please sign me up to the H2E listserve. My email address is:
For a complete list of materials produced by HCWH, visit our website at
www.noharm.org.
We’d like to know about any programs you have initiated in your facility, or any
mercury1 related concerns or needs you might have. Please feel free to use the
space below or attach/fax a separate sheet.
Without Harm
Fax back form to HCWH at 202-234-9121 or email info@hcwh.org for more
information.
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharin.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
The I (.1- certification mark and term ate the sole property vl the C liluhne Free
Products Association and arc only used by iiutlwrired and certified users.
Instruments,
Products, and
Laboratory
Chemicak
Used in
Hospitals
That May
Contain
•Mercury
This list should not be assumed to he
complete. You may want to check one
of the following resources for updates
and additions:
www.masco.org
www. n i h. gov/od /o rs/d s/no me r c u ry/
www.sustainablehospitals.org
Thermometers
n Body temperature thermometers
n Clerget sugar rest thermometers
n Heating and cooling system
thermometers
n Incubator/water bath thermometers
a Minimum/maximum thermometers
n National Institute of Standards and
Technology calibration thermometers
a Tapered bulb (armored)
thermometers
Sphygmomanometers
Gastrointestinal tubes
n Cantor tubes
a Esophageal dilators (bougie tubes)
n Feeding tubes
n Miller Abbott tubes
Dental amalgam
Pharmaceutical supplies
a Contact lens solutions and other
ophthalmic products containing
thimerosal or phenylmercuric nitrate
D Diuretics with mersalyl and
mercury salts
n Early pregnancy test kits with
mercury-containing preservative
n Merbromin/water solution
n Nasal spray with thimerosal,
phenylmercuric acerate or
phenylmercuric nitrate
0 Vaccines with thimerosal (primarily
in hemophilus, hepatitis, rabies,
tetanus, influenza, diphtheria and
pertussis vaccines)
n Cleaners and degreasers with mer
cury-contaminated caustic soda or
chlorine
Batteries (medical use)
n Alarms
n Blood analyzers
n Defibrillators
n Hearing aids
n Meters
n Monitors
D Pacemakers
° Pumps
° Scales
° Telemetry transmitters
n Ultrasound
D Ventilators
Batteries (non-medical uses)
Lamps
D Fluorescent
° Germicidal
Q High-intensity discharge
(high pressure sodium, mercury
vapor, metal halide)
° Ultraviolet
Electrical equipment
n Tilt switches
° Air flow/fan limit control
a Building security systems
n Chest freezer lids
a Fire alarm box switches
° Lap-top computer screen shut-off
n Pressure control (mounted on
bourdon tube or diaphragm)
n Silent light switches (single-pole
and three-way)
® Temperature control (mounted on
bimetal coil or attached to bulb
device)
° Washing machine (power shut off)
Float control
° Septic tanks
Q Sump pumps
Thermostats (non-digital)
Thermostat probes in electrical
equipment
Reed relays (low voltage, high
precision analytical equipment)
Plunger or displacement relays (high
current/high voltage applications)
Thermostat probes in gas appliances
(flame sensors, gas safety valves)
Pressure gauges
c Barometers
a Manometers
° Vacuum gauges
Other devices, such as personal computers, that utilize a printed wircboard
n Blood gas analyzer reference elec
trode (Radiometer brand)
c Cathode-ray oscilloscope
c DC watt hour meters (Duncan)
■ Electron microscope (mercury may
be used as a damper)
° Flow meters
° Generators
n Hitachi Chem Analyzer reagent
° Lead analyzer electrode (ESA
model 301 OB)
n Sequential Multi-Channel
Autoanalyzer (SMCA) AU 2000
■ Vibration meters
Laboratory Chemicals
That May Contain Mercury
(Compiled in 1997)
This list is intended to demonstrate the
wide variety of laboratory chemicals
that may contain mercury. It was
derived from examining the
Massachusetts Water Resources
Authority Mercury Source
Identification Program Database.
Some of the chemicals may contain
added mercury and others may contain
mercury’ as a contaminant in a feed
stock. If the mercury’ is a contaminant,
its presence or absence may vary’ from
lot to lot. In the case of kits, it is nec
essary' to consider separately each of
the reagents that make up the kit.
This list should not be assumed to be
complete. Request that vendors dis
close mercury' concentration on a
Certificate of Analysis for all chemicals
ordered.
°
n
□
°
n
□
n
°
a
a
a
a
o
°
n
a
°
D
a
□
□
n
°
a
°
n
a
a
a
°
a
a
°
°
n
■
Acetic acid
Ammonium reagent/Stone
analysis kit
Antibody test kits
Antigens
Antiserums
Buffers
Calibration kits
Calibrators
Chloride
Diluents
Enzyme Immunoassay test kits
Enzyme tracers
Ethanol
Extraction enzymes
Fixatives
Hematology reagents
Hormones
Immunoelectrophoresis reagents
Immunofixationphoresis reagents
Immu-sal
Liquid substrate con
Negative control kits
Phenobarbital reagent
Phenytoin reagent
Positive control kits
Potassium hydroxide
Pregnancy test kits
Rabbit serum
Shigella bacteria
Sodium hypochlorite
Stains
Standards
Sulfuric acid
Thimerosal
Tracer kits
Urine analysis reagents
Wash solutions
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit.
visit Health Care Without Harm on the Web at www.noharm.org.
The PCF certification mill .mJ term are the sole property of the Chlorine Free
l’n JiKts Association .mJ are only toed by auih.-n:cd and certified iners
Fart Sheet
What is the problem
with mercury fever
thermometers?
Very small amounts of mercury can do
significant damage. One gram of mer
cury is enough to contaminate all the
fish in a lake with a surface area of 20
acres. A typical mercury thermometer
contains approximately 0.7 grams of
mercury (700 milligrams), but larger
thermometers can contain as much as
three grams. Both short term and long
term exposure to mercury can cause
serious health problems for humans
and wildlife.
How toxic is mercury?
.
i publication is part of Going Green. A Resource Kit fo r Pollution Prevention in Health
I copies of this or other publications included in the kit, or to find out how to get a
visit Health Care Without Harm on the Web at w-ww.noharm.org
October 15, ?001
Mercury affects the nervous system
and can impair the way we hear, talk,
see, walk, feel and think. Humans are
exposed to mercury through contami
nated air, water or food or directly
through the skin. In fact, long before
we had scientific facts to prove mer
cury’s toxicity, there was evidence that
mercury poisoning resulted in nerve
damage. In the 1800’s hat makers
were exposed to mercury during the
wool felting process. The strange and
unpredictable behavior of Lewis
Carroll’s “Mad Hatter” in Alice of
Wonderland was a portrayal of hat
makers who had gone “mad” from mer
cury poisoning.
Does one broken
fever thermometer
really pose a health risk
to the consumer?
Yes, it can if not cleaned up properly.
Every year, there are 15,000 phone
calls to poison control centers about
broken mercury thermometers. When
a mercury thermometer breaks, rhe liq
uid silver-colored metal can spill onto
the floor or carpet. Breaking one fever
thermometer is unlikely to threaten
the health of the consumer if the
spilled mercury is cleaned up properly.
However, if the consumer fails to clean
up mercury either because he or she is
unaware that it has broken or because
it is difficult to gain access to the mer
cury (for instance because it has seeped
through a carpet), then the mercury
will eventually evaporate into the air
and reach dangerous levels in indoor
air. The risks increase if the consumer
attempts to clean up a mercury spill
with a vacuum cleaner, or if the mercu
ry is heated. The danger of significant
mercury exposure is greatest in a small,
poorly ventilated room.
Actual Case Studies1
In one case, exposure resulted
when 1.1 grams of mercury from a
broken fever thermometer were
collected and placed in a pan that
was laid on a hot kitchen stove. As
a result, the mercury vaporized
quickly. Two elderly patients devel
oped severe pulmonary edema,
diarrhea, confusion, tremors, and
coma, and died after 7 and 17 days
of hospitalization. A third patient
developed a skin rash that cleared
up after 3 weeks.
a Another case involved a 32 monthold girl who was afflicted by hyper
tension, irregular heartbeat, apathy,
irritability, excessive sweating and
acrodynia as the result of exposure
to mercury spilled from a broken
thermometer onto carpet. Three
months of treatment were required
before her condition improved.
n Three children, ranging in ages
from 20 months to six years old,
were exposed to mercury' from a
thermometer that had been spilled
on a carpet. They developed symp
toms including loss of appetite and
weight loss; sensitivity to light;
pink, sweating, and scaling palms;
eczema and itching. The two more
severely affected children required
four months of therapy before com
plete recovery.
°
Do fever thermometers
really contain enough
mercury to affect the
environment?
Yes. If you dispose of a mercury' ther
mometer in your regular garbage and
that trash is burned in an incinerator,
mercury vapors will be released into
the air. Mercury from landfilled
garbage can seep into groundwater or
can be released into the air as a toxic
vapor. Airborne mercury’ eventually
falls to earth, often into rivers and
lakes, where microorganisms transform
the mercury’ into a highly toxic form
called methylmercury’. Methylmercury’
builds up in aquatic animals, including
fish. It accumulates in muscle tissue,
and so, unlike some other pollutants, it
cannot be trimmed away when cooking
the fish. Mercury poses the greatest
threat to people who eat large amounts
of contaminated fish. For pregnant
women, eating contaminated fish poses
a special risk because mercury crosses
the placenta into the developing child.
While the amount of mercury’ in an
individual thermometer may seem
small, the total amount contained in
thermometers is significant. The
United States Environmental
Protection Agency considers mercury
thermometers one of the largest
sources of mercury to the solid waste
stream, estimated at 17 tons per year.
Clearly, thermometers are a meaningful
source of mercury to the environment
that can be easily reduced by switching
to non-mercury thermometers.
What happens if a
mercury fever thermometer
breaks in a child's mouth?
It is also common for children to break
fever thermometers in their mouths.
Mercury’ that is swallowed poses a low
risk in comparison with the risk of
breathing mercury’ vapor. The mercury'
passes that through the body and is
minimally absorbed, but it will contaminate the environment when it enters
the waste water system.
These include:
a
a
□
Digital electronic thermometers
Glass gallium-indium-tin (galinstan) thermometer
Flexible forehead and ear canal
thermometers
A recent statement by the American
Medical Association indicated that
non-mercury’ fever thermometers are
adequate diagnostic tools.
What are the risks that an
alternative thermometer
could poison the user?
There is no known or anticipated risk.
What are the environmen
tal consequences of nonmercury thermometers?
The known environmental damages
caused by alternative thermometers are
significantly less than those presented
by mercury thermometers. The primary’
environmental concern arising from use
of alternative thermometers relates to
the disposal of button cell batteries
used in digital electronic or ear canal
thermometers. Button cell batteries
used in digital thermometers contain
significantly less mercury' than a mercu
ry' thermometer—roughly 3.5 to 11 mil
ligrams of mercury’ per battery’.
When a mercury’ thermometer or a
button cell battery is thrown away and
burned in an incinerator, much of the
mercury' that it contains is likely to be
emitted to the atmosphere. However,
a mercury’ thermometer that breaks in
the home, or that breaks in the solid
waste system prior to burial in a land
fill, will release significantly more of its
mercury' than will a button cell battery'.
Notes
What are the alternatives
to mercury thermometers?
Several types of non-mercury’ ther
mometers are available commercially.
1. See Environmental Protection
Agency website: www.epa.gov/
glnpo/bnsdocs/hg/thermafaq.html
Without Harm
1755 S Street, NW
Suite 6B
Washington, DC 20009
Phone: 202.234.0091
Fax: 202.234.9121
www.noharm.org
info@hcwh.org
This publication is part of Going Green: A Resource Kit for Pollution
Prevention in Health Care. For additional copies of this or other pub
lications included in the kit, or to find out how to get a complete kit,
visit Health Care Without Harm on the Web at www.noharm.org.
fiuBUOvt.h*.
SOY INK
Thc PCF certification mark and tcnii arc the sole property of the Chlonne Free
Prcducu Association and arc only u.vJ by .hith.-rued and certified users.
ubM cv
e/icwiAA q eve/v
(kc/i^CH^vvete/v
L T H
7'
Hout
h
A Mercury Fever
Thermometer
Exchange is an
event at which
participants turn
in mercury fever
thermometers
brought from their
homes and, in
return, receive a
non-mercury fever
thermometer or
a voucher for an
ercury thermometers are made of glass the size of a straw,
with a silvery-white-liquid inside. Mercury fever thermometers
have been used for decades as a first step in caring for someone
^.l_who feels sick. But, ironically, the mercury thermometer can be
a risk to’the health of families and communities. Mercury is a toxic
substance that can harm both humans and wildlife.
Many families have had a mercury thermometer in their medicine chest
for years without breaking it. But mercury thermometers are very easy to
break and’veiy difficult to clean up. To function properly, mercury
thermometers must be “shaken down” before use, creating a constant high
potential for breakage. Public health officials across the country report a
steady stream (over 18,000 to poison control centers in 1998 alone) of
. concerned calls from broken mercury thermometers. Fever thermometers are
the largest single source of mercury discarded annually in municipal solid
waste, estimated at 17 tons of mercury per year.
alternative
thermometer.
■ j-
These exchanges
not only educate
about the
When a mercury thermometer breaks, ii is difficult to clean up properly.
Sometimes parents may not know that their child has broken a thermometer.
Sometimes mercury from the broken thermometer spills into a crack in the •
floor or soaks into a carpel. If mercury spills from a thermometer and is not
cleaned up, it will all evaporate, potentially reaching dangerous levels in
indoor air. A single broken fever thermometer, containing 0.5 to 1.5 grams
of mercury, is enough to. create a health risk if it evaporates into a small,
poorly ventilated room.
.
environmental
and public health
effects of mercury;
they also provide
participants with
the opportunity
to be part of the
solution to the
*
mercury problem.
Mercury affects the human brain, spinal cord, kidneys and liver.
It affects the ability to feel, see, taste and move. Il can cause tingling
sensations in the fingers and toes, a numb sensation around the mouth and
tunnel vision. Long-term exposure to mercury can result in symptoms that
get progressively worse and lead to personality changes, stupor and coma/
Wildlife populations, especially loons, are already exhibiting effects of
mercury poisoning. There is already so much mercury pollution that 39
states are currently warning residents not to eat certain species of fish
caught in all or some of the stale’s lakes, rivers, streams and coastal waters.
In pregnant women, mercury can pass through the placenta, where’it
affects fetal development by preventing the brain and nervous system from
developing normally. Affected children show lowered intelligence, impaired
hearing and-poor coordination. Their verbal and motor skills may be
delayed. Because of these threats to the developing fetus, the federal
government recommends that women who are pregnant or who may
become pregnant not eat mercury-contaminated fish.
th
INTRODUCTION
The success of an exchange or roundup depends on the successful
promotion of the event. If people do not know about the event, you’velost your opportunity to collect thermometers and to educate about the
health and environmental impacts of mercury. Your audience will define
the way you promote and plan your exchange, and therefore its budget
and ultimate success.
■
.
.
Experience has shown that there are three main types of exchanges,
based on the audience you are trying to engage. These exchanges are:
’ ,•
0 Hospital and Other Workplace Exchanges
e School Exchanges
•
.•
•
0 Community Exchanges
.
'
• •
. Workplace and school exchanges are relatively simple exchanges to
undertake. Primarily, this is because your audience is easily defined. You
have a few direct and straightforward means to inform possible participants
about the event. Promotion is therefore simple and inexpensive. In addition,
the audience is a “known quantity’’ and easily estimated..Based on either
number of employees or number of students, a simple’formula can then be
used to estimate the quantity of exchange thermometers or vouchers needed.
This helps simplify-budgeting.
•
*
•
•
Relative to workplace and school exchanges, community exchanges .
are more complex. You may need to use a wide variety of promotion
mechanisms including paid advertising. You may not be able to adequately
determine the demand for alternative thermometers or coupons and’be
required to adopt a “while supplies last" approach. Fundraising from a
broad range of potential sources may be required before you can hold a
community exchange.
.
•
•
•
Yet, all three types of exchanges can be highly successful events not
only in the number of thermometers collected, but in terms of environmental
and public health education. Like any project, their success is dependent on
good planning. This guide is intended not only to help encourage you to
undertake a thermometer exchange, but to help and assist you in the
process so that it is a success.
AN OPPORTUNITY
FOR EDUCATION
A mercury fever thermometer
exchange can provide a
wonderful opportunity to
educate about the hazards
of mercury. An exchange can
also inform people on the
actions they can take to
reduce the risk of mercury
contamination of the
environment and to protect
the health of their families
and the community. If you
host a staff exchange in your
workplace, it can be a part
of an employee wellness
program or a kick-off or
culmination event for an
in-house mercury
elimination program.
EASY TO COORDINATE
WITH BIG PAYOFFS
Mercury thermometer
exchanges are worth the
effort they take to put
together, since the benefits of
an exchange are numerous.
• When given information
about the hazards of mercury
thermometers, people are
eager to find a safe place to
get rid of them.
• Providing a free non-mercury
alternative is a big bonus.
People are always thankful to
get something for free,
especially when the associated
benefits are so positive.
• The public image and media
opportunities are sizable.
This Is a win-win situation for
everyone and the public and
the press will be receptive to
that message.
f
fact Vft
era
'TYVtI.
CHOOSING A
NON-MERCURY
FEVER
THERMOMETER
While there are a
variety of mercury-free
fever thermometers
available in the market
place, there are only
two main types that
fall within the budget
of an exchange. These
alternatives are the
Geratherm thermometer,
and the digital
thermometer. The
Geratherm functions
like, and is somewhat
similar to, a mercury
thermometer. Instead of
mercury, it contains
galinstan, an alloy of
gallium, tin and indium.
The digital thermometer
is electronic and uses a
button battery for power.
The table on the right
highlights some of their
benefits and drawbacks.
Geratherm *
Digital *
$4.00
Cost Estimate (1999) $2.80
Advantages Relatively inexpensive.
Easy to use and read.
Can have message
printed on case.
Disadvantages Poor data on long term
Somewhat expensive
Contains button battery
that should be recycled.
environmental impacts
of galinstan.
May be confused for a
mercury thermometer.
♦.See appendix B for contact information
REPLACEMENT OR VOUCHER
-------THERMOMETER
•
*
•
One decision that needs.to be made early in the planning process is
whether you want to distribute non-mercury thermometers, or work with
local retailers to accept vouchers for a free or discounted non-mercury
replacement Either option will work, but it is important to keep in mind
that the easier it is for a participant to exchange their thermometer for a new
one, the more likely they will participate in a program. Using a voucher may
be perceived as adding an extra step or hurdle. In addition, should you
decide to work with a retailer on a voucher program, it is important to
ensure that they do not sell mercury thermometers.
ESTIMATING HOW MANY
THERMOMETERS ARE NEEDED---------- ------ :--------------
Deciding how many thermometers are needed is important for
budgeting. As discussed earlier, the number of thermometers brought in is
directly related to promotion of the event. If only one mercury-free
thermometer is exchanged per household, a reliable estimate for workplace
and school events is easy to determine. For school roundups, experience has
determined an exchange rate range of 18% to 25% on a student population
basis, and for the workplace an exchange rate of 11%.to 18% based on
quantity of employees. If the event is well-promoted and you have good
support within the school or
Anywhere
Anywhere
workplace, you can estimate an
Location
Hospital
High School
exchange rate on the high end of the
Population
4000 employees 600 students
range given. For small schools and
Expected exchange rate
11% to 18%
workplaces (less than a population
18% to 25%
of 500), the return rate .is also typically
Level of exchange awareness
Low to -medium High
(guess-timate based on population size and level of promotion)
on the higher end of this range.
Because some participants in an
Estimated exchange rate
25%
13%
exchange bring in more than'one
Exchange factor
.13
.25
mercury thermometer, an extra level
of confidence is added in the estimate
Quantity of
4000 x .13= 390 600 x .25= 150
thermometers needed
of thermometers needed. This table
aMMEDWMHUanMMHM
gives two examples of how to estimate
the amount ot thermometers that will
be needed.
I
FUNDINGBefore you go looking for funding, it is important to know
what you are asking for. Is it money, thermometers, or other in-kind
support? The simplest exchange requires thermometers (or
vouchers), disposal, and perhaps some money for printed promo
tional materials. Compared to many programs, the funding budget
for a thermometer roundup can be rather small.
Your funding may also be helped if you have an opportunity to
piggyback an exchange with a community or children’s health fair, ’
an Earth Day event, or a local household hazardous waste collec
tion. If so. issues such as planning and publicity may be easier to
handle. It may be worth checking into your local community
calendar to see if such opportunities exist.
Some mercury exchanges can get quite involved, with high
profile names invited to attract media, or food at a reception to
attract participation. High-profile events will typically require a
higher budget While these niceties can turn the work of organizing
a simple exchange into a spiraling quest for.funding, they can also
provide excellent benefits. See the following section on receptions.
■ When looking for funding for your exchange, there are a variety of '
avenues that you can explore. Because mercury exchanges are a “feel good”
type of event, they are often well received by potential sponsors. Consider
both the public health and environmental aspects when soliciting funds.
Furthermore, funding may be in-kind. Do not ignore free food for a
reception, free disposal, and most importantly, free advertising or
promotional items. The following is a list of some sources that may be able
to help your fundraising.
. .,
POTENTIAL SPONSORS AND THEIR INVOLVEMENT
• Hospitals
• Corporate Sponsors
- A mercury7 exchange is a way for them
to be seen as environmentally responsible.
- It may fit with a corporate employee
wellness program.
- It may fit with a need to be seen as a
good community citizen.
•
•
• State or Local Pollution Control Agencies
In many areas of the country, mercury is increasingly
an area of regulatory7 concern. Your state or local
agency may have money targeted for mercury. .
• Pharmacy Chains/Thermometer Vendors
The larger pharmacy chains may be able to give
some form of discount program and/or free
thermometers. Some thermometer vendors have
given out complimentary thermometers.
• Solid and/or Hazardous Waste Haulers
Many ‘’haulers” have a financial interest in assuring their
loads are mercury-free. In addition, a mercury exchange
is an excellent way for this industry to promote itself as
environmental and community7 stewards,
In 1998, the American Hospital Association signed an
agreement to virtually7 eliminate their use of mercury7.
In 1999,.over 100 hospitals signed Health Care
Without Flarm’s pledge to practice mercury-free medi
cine. Your hospital may be interested in being
involved to help promote their mercury reduction
efforts, and promote their mission of community
health.
••
• Wastewater Treatment Plants
Wastewater treatment (WWT) plants have mercury
discharge permits that are becoming more stringent.
Some permits may have conditions, which allow the
\VWT plant to do pollution prevention work as a
means to meet their permit. A mercury thermometer
exchange is an excellent means to do, and educate
on, mercury7 pollution prevention.
• Household Hazardous Waste (HHW) Collection Facilities
' These facilities are in the business of collection of
HFIW and may7 be able to offer free disposal. Based
on their knowledge of mercury waste generators, they7 may
be able to offer good leads on related businesses in town.
RECEPTION--------7-- :------- ?-- —-- ;--- —
MERCURY
THERMOMETER
DISPOSAL
o
Experience has shown that (hose helping
with the exchange and those turning in their
thermometer will want to know tlie eventual
fate of tlie mercury in the thermometers.
Currently, die mercury in mercury
thermometers and other mercury-containing
devices is recycled using a process called
“roast, retort and distillation." Basically, the
mercury-containing items are crushed, and
heated so that the mercury evaporates and is
thus separated from the glass and other
debris. The gaseous mercury is then retorted
or condensed back to a liquid state. The
liquid mercury is then distilled to remove
impurities and can be used again in new,
mercury-containing products.
It may be a surprise to many that their
mercury may actually return back to
commerce in another mercury device.
While there is broad agreement on the
serious toxicity of mercury, and our
governments have called for its phase out, it
is still sold in products for which viable,
cost-competitive alternatives exist. This is yet
another reason why a mercury thermometer
exchange can be so important, for they can
be used to educate homeowners on other
places where mercury can be found in the
home and alternatives to its use.
To get a list of all facilities in your
area that can provide this process, contact
either your hazardous waste hauler, or your
state department of environmental
protection/services.
In choosing a facility; check references and
with state officials to insure the facility is
fully permitted and dial there are no EPA or
OSHA violations against tlie facility. This is
very’ important. So do your homework on
a facility’ before you send your mercury
to them.
Ask the mercury reclamation facility how
they want the mercury contained and labeled
for the collection, storage, and transportation
of the mercury thermometers. They will
probably want die thermometers collected in
a Department of Transportation (DOT)
approved shipping container. They should
lie able to provide you with this assistance.
A workplace reception can be a great way7 to help promote the
exchange, but at the same time can require a lot of planning time.
This must be weighed with the fact that a well-planned reception can
produce excellent benefits. High-visibility events often attract the interest
of senior management- As senior management gets involved in the
planning and implementation of the round-up, they gain a vested interest
in the long-term success of the mercury elimination program, not simply
as a one-time event organized by (he general staff. At a large urban
Boston hospital mercury-thermometer round-up reception, senior
management announced that they would design a program to eliminate ■
all mercury-containing blood pressure measuring devices and replace
them with non-mercury alternatives.
■
Benefits of High Visibility Exchange:
.
HCWH’s round-up at a Boston hospital focused on clinical fever
thermometers. Because of the success of this event, senior management
took it upon themselves to put a policy in place to round up all research
laboratory mercury thermometers and replace them with non-mercury
alternatives. The bottom line was that a larger event attracted the attention
of decision makers who bought into the program.
In the hospital setting, by7 virtue of- medical profession involvement,
a reception can help draw attention to mercury7 as a public health issue.
At a reception,’it is useful to have a display7 on mercury, mercury-free
alternatives in the home and workplace, andmercury pollution prevention literature.
Options for speakers include physicians or
other clinicians that can speak to the health
hazards of mercury, state or federal speakers
addressing the status of mercury’ legislation.
local environmental organization representa
tives and; workplace staff on what
that organization is doing
to address mercury
reduction/elimination.
MEMORIAL
HOSPITAL:
STRIVE TO BE
MERCURYFREE
BY 2003
IVH
Having at least cookies
and punch available during
the event is also a good idea.
One of the event’s goals is to
provide additional information
about mercury hazards.
Providing food, means that ’
people will stick around
and give you more time to
give them that information.
Providing cookies is also
celebratory7 and adds to
the positive atmosphere
of the event.
PUBLICITY
----------• -------------------------- -----------%
There are a variety of ways to publicize your exchange.
The following list provides some ideas on how to get the Word out.
(See Appendix A for templates)
• •
Workplace and School Exchanges
• Fliers (send home with students)
• Classroom of departmental meeting presentations by peers
• Table tents - cafeteria, lunch room or staff lounges
• Newsletters, including small neighborhood papers or “shoppers”
• E-mail announcements (the day before event, post an automatic
announcement "don’t forget your mercury thermometer tomorrow!”)
• Announcement in payroll checks
Community Events
• Fliers
• Public service television and radio spots
• Newspapers
• Community Websites
• Community, health and environmental
organizations' newsletters and e-mail listj
Event Coverage
• Contact radio (on-site coverage), newspapers
and television stations ahead of time to cover event.
• Submit articles to magazines, trade journals and community
organizations to summarize the event’s success.
EDUCATIONAL OPPORTUNITIES
•
•
Before the-event, collect enough educational materials to distribute.
Listed are just of sampling of information you might want to provide.
• HCWH’s Making Medicine Mercury-Free*
• HCWH’s Mercury Thermometers and Your Family’s Health*
(available in English or Spanish)
® HCWH’s How to do a Mercuiy Fever Thermometer Exchange*
• HCWH’s Mercury-free Thermometer Pharmacy Campaign information
• Your state fish advisories
• List of other mercury-containing items in. the home and workplace
• Local and state contact information about mercuiy disposal options
for other mercury-containing household items
* Please be sure to contact HCWH 3-4 weeks in advance with ’
■ an estimate of how many copies are needed
SAFETY AND
ENVIRONMENTAL
LOGISTICS
Il is important to make
sure that in all promotional
materials, participants are told
to bring in their thermometers
in a rigid container, and to
place the container in a
zip-lock bag, as a second
measure of protection. The
original case in which the
thermometer was bought
works perfectly, but any
other non-breakable container
(toothbrush case or plastic soft
drink bottle with screw-on top)
will also work. These precau
tions are important to prevent
the thermometer from breaking
on the way to the exchange,
and to protect the health and
safety of the participant, should
it still break.
ork with workplace or
stat Safety or hazardous
aterials specialists during
the event planning process
to ensure that regulatory and
compliance issues are being
considered. If you are going
to transport the collected
thermometers to the disposal
facility, it is important to
ensure that transport and
labeling regulations are being
followed. Mercury destined for
reclamation is considered a
federally designated “universal
waste”, but contact your state’s
hazardous materials section
to ensure you will be in
compliance with your stale’s
environmental requirements.
Someone with mercuiy
spill cleanup training, and a
mercury spill kit should be
available al the event.
LOCATION AND SCHEDULE OF THE-EXCHANGE —
••
It. is important to time the event of your exchange so that it is
convenient for those participating in the exchange.
In a workplace with shift workers, try at a minimum to schedule the
exchange over one shift change. It is easy for exchange participants to turn
in their thermometer- at the beginning or end of their shift. If possible, try to
hold the exchange for al least two hours, but longer is preferable.
The longer it is open, the greater the chance that word of mouth will remind
workplace staff of the event. For school events, make sure the exchange
occurs when students are able to participate. Before school starts and during
lunch are times that have proved successful, for school exchanges. •
Holding the exchange in a popular community meeting area will also
help the success of the exchange. Typically, the most successful meeting
place is the work or school cafeteria. Setting up the “exchange table”
outside the cafeteria doors will guarantee a steady stream of people.
In many hospitals, there is a shift change at the lunch hour. Accounting for
location and timing will help the exchange tremendously.
•
9
•
f
•
OTHER CONSIDERATIONS------------ :------- ---- :-----------•
•
Establish Guidelines
Many exchange participants will bring in more than one mercury
thermometer from their home. Make sure people understand that they can
bring in as many as they have. At the same time, it is important to have a
clear guideline on how many non-mercury thermometers they can take
home. Many exchanges have adopted a policy of one mercury- free
alternative per family. This way, participants aren't discouraged from
bringing in more than one thermometer, and are encouraged to collect
those from other family households.
•
•
•
•
•
It is also important to have a policy on accepting other
mercury-containing equipment. Unless you want to be inundated by
mercury-containing devices, never advertise your collection as anything
but a thermometer exchange. At the same time, it is hard to turn away
someone trying to do the right thing by bringing in their thermostat or
other mercury-containing device. Thermometer exchanges have accepted a
five-pound bottle of mercury, thermostats and other mercury devices. The
choice is yours, but having a good guideline in place before the day of your
exchange can save you lots of headaches.
•
Mercury from Home or business ■
•
’
On a related note, some workplaces will ask whether they can dispose
of some of their- business waste through your exchange program. Il can be
difficult when a representative from a department you have not worked
with asks for a special favor, hoping to save on mercury disposal costs' It is
especially difficult if you do not work in the workplace and get caught in a
struggle between that department and the one helping facilitate the
exchange. Again, the choice is yours to make. What you should be aware of
is that many states regulate business waste differently than household waste.
If you are deliberating whether to take workplace mercury through your
progiam, it is advisable to check with your slate regulators, and/or disposal
facility to see if you will be violating any state laws. You should be prepared
when the science department head asks if you could take the sixty broken
lab thermometers they have been storing, or the hospital maintenance
department asks you to take in fifteen pounds of mercury.
Plan for Success
Either due to good promotion or a small budget you should also be.
pi epared with a contingency plan in case you run out of thermometers.
Will you offer a voucher that the participant can redeem in the future, will
you turn them away, or do you promote the exchange of free thermometer
only ’‘while quantities last”?
It is also good to be prepared tor people wanting to exchange a
thermometer the day after the event, or after the exchange has closed for
the day. There will typically be employees that will be reminded of the
exchange only when they see the exchange table on the day of the event.
To collect from these latecomers, you can hold and advertise subsequent
make-up days, or in the case of a workplace exchange, provide the lime and
office or other location where they can drop them off during working hours.
This is particularly important where the workplace has three shifts. Il may be
logistically too difficult to hold the exchange during the second shift change.
Providing the third shift with an option not only makes the exchange
inclusive but also more successful. Finally, make sure to make participants
aware of the hours and location of the local household hazardous waste
facility. They might have mercury or other wastes that they want to get
rid of responsibly.
Close the Loop
Some slates .such as Minnesota, have banned.the distribution of mercury
thermometers by hospitals, to new parents. The loophole that still exists is
that most pharmacies still sell mercury basal and fever thermometers. If you
have the time and resources, send letters to all your local pharmacies and
ask them to stop selling mercury thermometers, once those still in slock are
sold. Follow your letter with a phone call, about one week after you have
mailed the letter. (See Appendix C for a sample letter). This is especially
important if you are doing an exchange in a hospital. It can be very
embarrassing to your efforts if a reporter covering the event inquires at the
hospital pharmacy and finds them still selling mercury7 thermometers.
Finally, educate yourself and have information available, which can
answer questions raised on the accuracy of mercury alternatives.
(See Appendix D for more information:)
COLLECTION PROCEDUREParticipants remove unbroken thermometer from their rigid container
and place it on the piece of plastic (or thin bubble wrap) which is spread
on a tray. If thermometer is broken, place back in rigid container and place
in large collection container.’
Keep track of number of thermometers collected and number of families
participating (which should be the same as the number of non-mercury
thermometers exchanged). A flip chart may be used to visually show
progress throughout exchange event.
When tray "fills up”, wrap slack of unbroken thermometers in. plastic,
wrap with rubberband or tape, and place “wad’.' in collection container.
The collection container should be labeled “Mercury Thermometers” and
have a lid (five gallon containers used in food service or for drywall spackle
work well.) Participants should not reach in or place their thermometers
directly into container.
Broken thermometers should be placed directly into collection container
EVENT DAY CHECKLIST
Table and chairs for volunteers
Banner/posters (see appendix A)
Mercury spill kit and emergency procedure in place
Collection tray .
Plastic wrap or thin bubble wrap
Collection bucket with lid
Sign "Mercury Thermometers Collection" on bucket
Large trash can for rigid thermometer holders
Flip chart, marker - tracking participation
Media,'public affairs readiness
If Holding a Reception:
I
Number of tables, table cloths .
Vendor accommodations
Speaker podium. PA system
Food, drinks and trash containers
—
-
HOW CAN 8 PREVENT
MERCURY POLLUTION?
(HCWH does not endorse any
of the following vendors)
Improper mercury disposal includes pouring
it down drains, putting it in the trash, '
and burning it. in barrels and incinerators.
Digital Thermometers
HERE'S WHAT YOU CANDO:
Know which products contain mercury--.
» • Becton
—•V » . 0‘S ’ ' - ■ ■ •Dickenson
* - r “ . » <“ 1 —"■ —' - •> * •
1 Becton Drive
Franklin Lakes, NJ 07417
201-847-6800
www.bd.com
Avoid buying products that contain
mercury whenever rion-mercury
substitutes are available.
Recycle mercuiy-containing products
through Household Hazardous Waste (HHW)
collections in your area. (Call your town
office for more information).
•
£ C
•
Conserve energy to reduce reliance on coal
burning for fuel, which is a major source of
mercury pollution.
PolyMedica Corporation
11 State Street
Woburn, MA 01801
781-933-2020
www.polymedica.com
Omron Healthcare,Inc.
L
_____
MAKING MEDICINE MERCURY-FREE
300 Lake View Parkway
Vernon Hills, IL 60061
800-231-3434
www.omronhealthcare.com
MERCURYTHERMOMETER SWAP
|Geratherm
Thermometer
EARTH day celebration
. ■
THURSDAY, APRIL 22
HOSPITAL ROTUNDA, llam-4pm*
R.G. Enterprises
• Hospital Staff - bring in your home mercuiy
thermometer for a free digital non-mereury
thermometer.
‘
. ■
® Reception at noon. Speakers from (your hospital)
community, invited speakers, and Health Care
Without Harm.
• Find out more about- mercuiy — what your hospital is
doing to eliminate its use and what you can-do in your
home to minimize/eliminate its hazards. •
--A. >
2000 Town Center, Suite 1900
Southfield, MI 48075
800-992-9497
email rgenterprises@msn.com
V
I
* 3rd shift - go by Housekeeping Office, 6:30am, April 22.
- . kA* .
* Available electronically at www.noharm.org
_a . '
MERCURY CONTAINING PRODUCTS
ALTERNATIVES/EXAMPLES
Thermometers
Digital thermometers (don’t forget to recycle the battery)
Batteries
Alkaline batteries (look for mercury-free batteries, dispose
of others at local HHW collections)
Thermostats, switches
Electronic or mechanical devices - dispose of the mercurycontaining items at local HHW collections
Contact lens solution
Solution without Thimerosal - check ingredients
Light bulbs (fluorescent, mercury vapor, neon,
metal halide, hp sodium)
Tungsten Filament (dispose of mercury-containing
lightbulbs at local HHW collections)
Soaps (including antibacterial soap)
Soaps without Triclosan
Detergents/cleaners/bleach: Clorox
Clorox Plus® (not other Clorox®), Austin-
|V\A
THE CASE AGAINST MERCURY:
THE PROBLEM WITH
MERCURY THERMOMETERS
Mercury causes a variety of health effects,
particularly for young children, including
nervous system damage, liver damage, kidney
damage, muscle tremors, impaired coordination,
and menial disturbances.
• A thermometer contains about 0.5-1.5 grams of
mercury-. One gram of mercury can contaminate a
rweny-acre lake with enough mercury to cause
public advisories (warnings) to limit consumption
of fish caught in that lake.
• Fever thermometers are the source of 17 tons
of.mercury discarded annually in the municipal
solid waste!.
• Eliminating even small amounts of mercury has a
beneficial effect on the environment, and reduces
the potential for human mercury poisoning.
• (Your hospital) is committed to eliminating
non-essential uses of mercury and mercurycontaining products. Removing mercury
thermometers is a responsible action in continuing
to serve the health care needs of our communities
while protecting the environment. Digital
thermometers provide comparable accuracy
and do not compromise patient care in any way.
•
•
• Thank you to Acme Technologies Inc. A Waste Management Company for the donation
■ of the digital thermometers for this event!
For more information contact the Office of Safety
and Environmental Programs, (phone number).
_
—
RADIO AD .
Radio Script for Mercury
Thermometer Exchange
- 30 second spots - .
Many families have had a mercury
thermometer in their medicine chest for
years, without it breaking. Yet, public
health officials warn that a broken
thermometer can pose a serious risk
to your family’s health and the
environment.- Even if you’ve never
broken a mercury thermometer, it
doesn’t mean you never will.
Protect your family’s health - Bring your
mercury thermometer to (collection
facility) before it breaks and receive a
non-mercury replacement free while'
supplies last'.
For more information, call
(your organization-and phone number).
Your address here
Dear Pharmacy,
As you may know, mercury has been identified as a major source of
environmental pollution. It is also widely recognized that elemental mercury
. and mercury compounds are hazardous to human health. Because of these
concerns, there are now many voluntary and legislative initiatives around the
country aimed at eliminating the use of products containing elemental mercury
or mercury compounds.
In Spring 1999, USA Today ran an article entitled, “Mercury Thermometers
Fall Out of Favor”. This article highlighted the concerns with mercury,
numerous programs around the country collecting mercury-thermometers, and
mercury-free thermometer promotions. Some states, such as Minnesota, have
banned the distribution of mercury-containing thermometers to new parents by
hospitals. Across the country, communities and organizations are holding
mercury thermometer exchanges or take-back initiatives.
Yet there remains one large problem with these mercury thermometer
initiatives. Many pharmacies still sell mercury thermometers. To close this
loophole we are writing to invite you to be a voluntary partner in (your
program). To become a partner, we are asking that you sign on to the national
Mercury-free Thermometer Pharmacy (MTP) campaign sponsored by Health .
Care Without Harm (HCWH). HCWH is an international campaign with over 250
participating organizations, including over 70 hospitals and other health-based
organizations such as the American Nurses Association, and the American Public
Health Association. The campaign is focused on transforming the healthcare
industry so that it is no longer a source of environmental harm by eliminating
pollution in healthcare practices. ■■
•
The Mercury-free Thermometer Pharmacy (MTP) campaign is aimed at
promoting those pharmacies that practice mercury pollution prevention. All that
is required to become a MTP is a written commitment, on your letterhead, to .
voluntarily discontinue the sale of mercury basal and fever thermometers once
those in stock are sold (a sample letter is enclosed). In turn, MTPs will be.
promoted nationally on the HCWH website (www.noharm.org).
We hope that you take this opportunity to be a community leader in
pollution prevention by signing on to the MTP campaign. Please find enclosed'a
copy of HCWITs Mercury Thermometer educational brochure. Should you have
further questions please do not hesitate to contact me at (phone number).
Sincerely,
!_____
—
------------------------------------------------------- ----------------------
------ ---------------------- ------------------------- - ------- - -------- .
* Available electronically at www.noharm.org
—
■
■
PHARMACY LETTERHEAD
HOT-i
P.O. Box 6806
Falls’ Church, VA 22040 ■ ’ -
.
■
..
.
Dear HCWH, •
Our Pharmacy has recognized that mercury has been
identified as a major source of pollution. We are also aware
that elemental mercury and mercury compounds are known
to be hazardous to human health and the environment.
We support the efforts of Health Care Without Harm, the
American Hospital Association (AHA) and the Environmental
Protection Agency (EPA) calling on hospitals to reduce the
volume and toxicity of their waste, specifically eliminating the
use of mercury.
• ’
We recognize that mercury basal and fever thermometers
■if used incorrectly, or broken, may contribute mercury to the
environment: We also recognize that cost-effective mercuryfree alternatives to these products exist for our customers.
We are therefore pleased to become a Mercury-free.
Thermometer Pharmacy (MTP). In becoming a MTP, we commit
to ending the sale of both basal and mercury thermometers once
our current inventory has been sold. Please add our name to
your national list of pharmacies that have discontinued the sale
of mercury-containing thermometers.
%
• •
Sincerely,
•
•(
SELECTING NON-MERCURY FEVERTHERMOMETERS
•
•
•
*
•
Alternatives to glass, mercury thermometers are quite appealing as they are easier and faster to use and avoid the shortcomings
of. glass mercury' thermometers. 7he risks of broken glass and exposure to'mercury are eliminated, as well as the cost of a clean-up .
and disposal of mercury' horn a broken thermometer. With the variety of alternatives available, it is essential that one make an
educated choice, to ensure that the tool satisfies the task. Here are some points worth thinking about when you consider ther- .
mometers:
'
•
•
*
a
*
•
•
I.ACCEPTABLE STANDARDS OF ACCURACY
Thermometers for medical use are typically tested to voluntary standards set by the American Society of Testing and Materials
(AS77M)1. The following table shows the maximum error allowed. One sees that glass/mercury' and electronic thermometers have
the same requirements over the range, of 96.4 - 106 F.
’ .
•
•
Maximum Error over Temperature Range Shown
•
* •
Thermometer Type
ASTM Procedure!
<96.4 F ’
96.4 < 98 0 F
.98.0 -102 0 F
Mercuiv in Glass
E667-861
(reapproved
1991)
*•
+ 0.4
+ 03
+ 0.2.
•
•
•
>102 -106 F
>106F
+ 0.3. ‘
+ 0.4
•
+ 0.3 ’
Electronic Thermometers Ell 12-86 1
+ 0.2
t 0.5
+ 0.5
+ 0.3
(reapproved 1991)
It is important to note that many thermometers read out to a smaller division than the accuracy of the thermometer itself.
For example, digital thermometers which read to 0.1 degrees F may be accurate only to + 0.2 F or less. If the accuracy is
+ 0.2 degrees F. the true terhperature of a thermometer reading 98.9 F is in the range of 98 7 - 99-1 degrees Fahrenheit.
Therefore when selecting a thermometer, one must look closely at the accuracy, rather than the smallest increment reported
2. ACCURACY OF GLASS/MERCURYTHERMOMETERS
Inherent in any discussion of alternatives is the assumption that glass/mercury thermometers are accurate. Data suggests that our
faith in. glass/mercury thermometers' may be misplaced.
.
•
Leick-Rude and Bloom- describe a study in which axillary temperature in neonates was taken with non-mercury thermometers
and compared with it “standard ’ of glass/mercuiy thermometers. For the purpose of the study, the accuracy of each glass/mercury'
thermometer was tested as a condition of accepting it for the study. 25% of the glass/mercury thermometers tested differed from
the reference thermometer by >0.2 degrees Centigrade and were deemed unacceptable for use in the study The authors cite
another study in which 28% of glass/mercury- thermometers were discarded because they differed by more than 0.1 degree
Centigrade from the reference thermometer. The authors raise concern as to the accuracy of glass/mercury thermometers for
•
general use. when one out of four of those tested was not deemed accurate enough. (In fact, the ASTM standard for glass/mercury ' '
medical thermometers specifies a maximum allowable error of + 0.1 C in the cited range).
3. FAVORING THE OLD STANDARD
Chamberlain and Terndrup' remind us that “Whenever a new clinical test is introduced, investigators measure its accuracy by
| comparing it to an accepted standard, termed rhe 'gold standard’. Because of this comparison to the old standard, initial testing will,
* by definition, favor the old method, even if the new clinical test is a better lest .
4. USE OF RECTAL, ORAL, OR AXILLARY READINGS AS A REFERENCE FOR TYMPANIC TEMPERATURE
• The publication “The Clinical Utility of Ear Thermometers'” describes different methods.and their limitations for measuring body
• temperature It cites that the medically accepted “gold standard" for core temperature is pulmonary artery blood temperature.
However this is an invasive technique, so rectal, oral, or axillary readings are often used as a crude estimate of body core tempera
ture. Each site’is reflective of a different blood supply, with separate rates of change with a rising or falling body temperature.
Additionally each site has variables unique to that site that influence the body temperature measured. The publication concludes
that since each site provides its own characteristic temperature properties, comparing a tympanic temperature directly with oral,
axillary, or rectal temperatures is inherently-flawed
.
.
The lesson here is that with an understanding of how tympanic thermometers work, they offer a sale, convenient alternative to
oral, axillary', or rectal temperature measurement. Education is critical to satisfactory performance, and manufacturers are well
prepared to advise and coach clinicians on the use of their products.
5. CUSTOMER SATISFACTION
t
.
. ,
,
. , ,
.
Numerous interviews with users of non-mercury thermometers provide convincing evidence that alternatives are viable and wellreceived in health care facilities.-
REFERENCES:
1) 1997 Annual Book of ASTM Standards. Roberta A. Storer. Editorial Services Director. American Society of Testing and Materials (ASTM). West Conshohocken. PA
2) MK Leick-Rude and Bloom I.F.
comparison of temperature-taking methods in neonates". Neonatal Network; August. 1998, Volume 17 No. 5. pp. 21-37.
3) James M. Chamberlain. MD, and Thomas E. Terndrup, MD, ’New light on ear thermometer readings" Contemporary Pediatrics; March. 199-i4) The Clinical Utility of Ear Thermometer, Published by Braun Thermosean. Pub. No. 0996-267P-R1097
Provided by the Sustainable Hospitals Project
A project within the Lowell Center for Sustainable Production-at the University of Massachusetts Lowell, providing
technical support to health care facilities. Visit our website: w^vaimLedu/centers/LCSP/hospitals/ or contact us at shp@uinl.edu
or 978-934-3386 for more information.
• .
>
-
....
r- ■
■
•
•■...■;■
.-.
■
.
$
■
■
-
■ .
■
■■■
■
■
■
■
■
v
' ■
'
.
s
i<
yr?
'
A'
J
-
•
.
.■
■'
-U
; ;■
.< ■
/*;•
• ; * •
: ■ ,
£ t? ©
-
- •
•:
■
•
/
■
: ru': '
*
TH CARE WITHOUT HARM
703-237-2249 • FAX: 703-237-8389 •
-
■
• P.O. BOX 6806 • FALLS CHURCH, VA 22040
EMAIL: NOHARMSU1P.ORG • WWW.NOHARM. ORG
•
* *75*
pXL.^ ^Xfcf tz-^^-la2j^A «3X
.
Position: 246 (69 views)